Calin Pop1,2, Oana Florentina Gheorghe Fronea3,4, Lavinia Pop1, Adriana Iosip1, Lucian Dorobantu5, Coralia Cotoraci2, Cornelia Bala6, Dana Pop7, Maria Dorobantu3,4
1 Emergency Clinical County Hospital, Baia Mare, Romania
2 “Vasile Goldis” University, Faculty of Medicine, Arad, Romania
3 Department of Cardiology, Clinical Emergency Hospital, „Carol Davila”
4 University of Medicine and Pharmacy, Bucharest, Romania
5 Monza Hospital, Bucharest, Romania
6 Department of Diabetes and Nutrition, „Iuliu Hatieganu” University of Medicine and Pharmacy, Cluj-Napoca, Romania
Abstract: Objectives – To estimate the prevalence of high normal blood pressure (HNBP) and to find if subjects with HNBP have more often other cardiovascular risk factors. Methods – A representative sample of 1970 Romanian adults was enrolled in SEPHAR III survey. Blood pressure measurements were performed according to current guidelines and all subjects were evaluated by a 71-item survey questionnaire together with extensive evaluation for target organ damage. Results – Prevalence of HNBP was 11% [45.1% high blood pressure (HBP), 43.9% normal blood pressure (NBP)]. Values of weight, waist circumference, body mass index, total and LDL cholesterol, triglycerides, fasting blood glucose, glycosylated hemoglobin – HbA1c, uric acid, serum creatinine, glomerular filtration rate estimate by CKD-EPI Equation, albumin/creati-nine ratio, intimae-media thickness, rates of arterial stiffness and diastolic dysfunction, left ventricular mass, interventricular septum and posterior left ventricle wall thickness, left atrial volume and dilatation were significantly highest in HNBP sub-jects than in NBP. Conclusions – Subjects with HNBP represent ~11% of the population and most of thems had an elevated cardiovascular risk. It’s essential to educate the general public and health care providers to be aware of these individuals and of steps that should be taken to treat modifiable cardiovascular risk factors.
Keywords: high normal blood pressure, prevalence, target organ damage, cardiovascular risk factors, high blood pressure.
INTRODUCTION
In 2017, US guidelines developed jointly by the Ameri-can College of Cardiology (ACC), American Heart Associa-tion (AHA), and other societies classify the 130 to 139 mmHg /80 to 89 mm Hg range as stage 1 high blood pressure (HBP)1. For the same blood pressure (BP) values, the former 2013 and the new 2018 European Society of Hypertension’s (ESH) / European Society of Car-diology (ESC) Guidelines for the management of arte-rial hypertension consider those with 120–129 mmHg and/or 80–84 mmHg to have normal blood pressure (NBP) and those with 130–139 mmHg and/or 85–89 mmHg to have high normal blood pressure (HNBP), with the intent to alert patients and physicians to pro-vide lifestyle education and sometimes medications2-7.
Romania, as previously shown in the three-national representative surveys [SEPHAR I (2005), SEPHAR II (2012)] and SEPHAR III (2016) – Study for the Evalua-tion of Prevalence of Hypertension and Cardiovascu-lar Risk in Romania], is a high cardiovascular risk East European country with a high prevalence of general HBP around 45.1%8-11.
Furthermore, with this study, we aimed to know the prevalence of HNBP and to find if these subjects have more often other cardiovascular risk factors than normotensives in order to provide a basis for preven-tives strategies for HBP and CVD.
METHODS
Detailed SEPHAR III methodology has been previous-ly published; therefore, we briefly present below only those aspects regarding the collection of SEPHAR III data that are the object of this study10,11
- SEPHAR III: sample selection and data collection
The SEPHAR III survey was conducted beetwen 2015/2016 in two stages and for an adult Romanian po-pulation of 16,269,839 adult citizens, of which 40.41% are estimated to be hypertensive patients (based on SEPHAR II results), with a maximum error of 2.18% at a confidence level of 95%, the minimum required sam-ple size was 1379 study participants8. During the two study visits, scheduled at a 4-day interval, all enrolled individuals were evaluated by the following: 71-item survey questionnaire, anthropometric, and BP measu-rements, together with investigations for target organ damage, blood, and urine sample collection after pro-per fasting time (8–14h prior).
- Blood pressure measurement
BP measurement technique and defi nitions of hyper-tensions were in line with 2013 ESH/ESC Guidelines2.
- Diagnostic Criteria
The classification of optimal, NHB, HNBP and HBP were done in accordance with the 2013 ESH/ESC Gui-delines, unchanged by the 2018 ESH/ESC new release Guidelines2,3. The BP category is defined by the highest level of BP, whether systolic or diastolic.
A. Optimal and normal BP – NBP, defined as not being on antihypertensive medication and having for optimal BP an SBP <120 mmHg and/or DBP <80 mmHg, and for normal BP an SBP 120–129 mmHg and/or DBP 80–84mmHg.
B. High normal BP – HNBP, defi ned as not being on antihypertensive medication and having an SBP of 130–139 mmHg and/or DBP of 85–89mm Hg: Ex, 136/70 mmHg was classified as HNBP but 136/90 mmHg as HBP, 126/70 mmHg was classified as NBP but 126/85 mmHg as HNBP.
C. Hypertension or high BP– HBP, defined as SBP at least 140 mmHg and/or DBP at least 90mmHg at both study visits, using the arithmetic mean of the second and third BP measurement of each study visit (without taking into consideration the first BP measurement from either visit), or pre-viously diagnosed hypertension under treatment during the previous 2 weeks, regardless of BP va-lues.
D. Controlled BP values were defined as SBP less than 140 mmHg and DBP less than 90 mmHg in treated hypertensive patients.
- Risk factors and diagnostic categories Detailed SEPHAR III data collection for risk factors and diagnostic categories has been previously publi-shed10,11. The use of the special medical caravan – SEPHAR BUS – has facilitated the fieldwork of the investigators and for the first time allowed them to perform a complete evaluation of target organ damage in a large number of subjects in a relatively short time interval.
- Cardiovascular risk classification
Total CV risk estimation was done using SCORE risk estimation system recommended for adults > 40 years of age, unless they are automatically categorized as be-ing at high or very high-risk, based on documented CVD, DM (> 40 years of age), kidney disease or highly elevated single risk factor. We use charts for high risk countries, as recommended for Romania in the 2016
edition of ESC cardiovascular disease prevention gui-delines7.
STATISTICAL ANALYSES
Statistical analysis was performed with IBM SPSS Sta-tistics 20.0 software at a significance level of p ≤0.05. A descriptive analysis (means, medians, standard de-viation and range for continuous data and frequency analysis for categorical data) was performed for all the target variables. Kolmogorov-Smirnov test was used to analyse continuous data distribution, according to which appropriate tests were further used in analysis: independent samples t-test or Mann-Whitney U test for differences between means of 2 independent groups, and ANOVA or Kruskal-Wallis test for diffe-rences between means of 3 independent groups. Chi-square test was used to analyse differences between categorical data. Binary multiple logistic regression using a stepwise likelihood ratio method including multicollinearity testing (tolerance less than 0.1 and VIF value greater than 10) was used for validation of predictors of HNBP and HBP (as dependent variable). Variables for which statistically significant differences between the 3 study subgroups were highlighted were used as independent variables (predictors) in regres-sion analysis. Data was weighted for region, locality type, age groups and gender.
RESULTS
- Prevalence of NBP, HNBP and HBP
A total of 1970 subjects were involved in statistical analysis: 1034 were females (52.4%) and 936 males (47.6%), mean age 48.5±17.5 years.
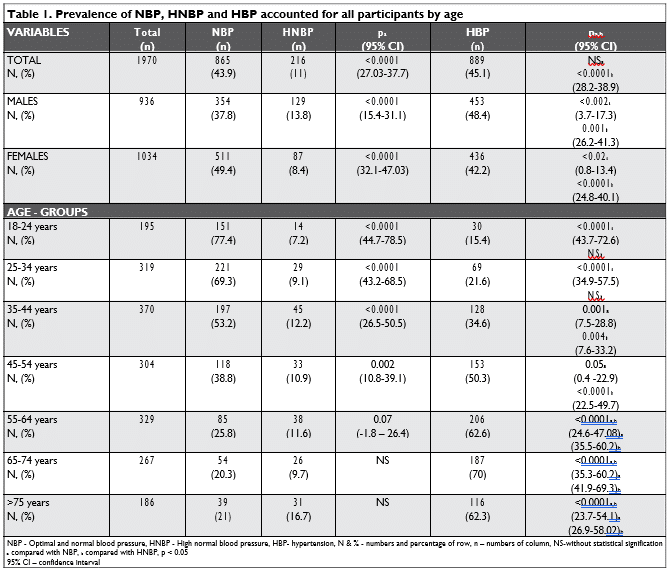
Categorized by blood pressure status, 865 (43.9%) subjects had NBP, 216 (11%) subjects had HNBP and 889 (45.1%) subjects had HBP. Individuals with HBP were older (mean age 55.7 ±15.6 years) than those with HNBP (mean age 51.1 ±17.1 years) and NBP (mean age 40.5±15.9 years), p<0.0001 (95% CI 18-85, respectively 18-91) – Table 1.
Gender prevalence for HNBP was 13.8% in males and 8.4% in females (p = NS, non-signifi cative) and 48.4% vs 42.2% (p=0.06) for HBP individuals. HNBP prevalence is increasing across age groups from 7.2 in the 18–24 years group up to 12.2 % in the 35-44 years’ group and then decreased with increasing age, except for those who were in the more than 75 years’ group. Subdividing the population by age and gender showed that in males the prevalence of HNBP peaked at an age of 25–34 years and in females at an age of 35–44 years (Figure 1).
As expected, HBP prevalence is increasing indepen-dent of gender across age groups, from 15.4% in the 18–24 years group up to 70 % in more than 65 years’ group (Table 1, Figure 1).
Global rate of HBP awareness accounting for a rate of 80.9%.
- Characteristics of study groups categorized by gender and blood pressure status.
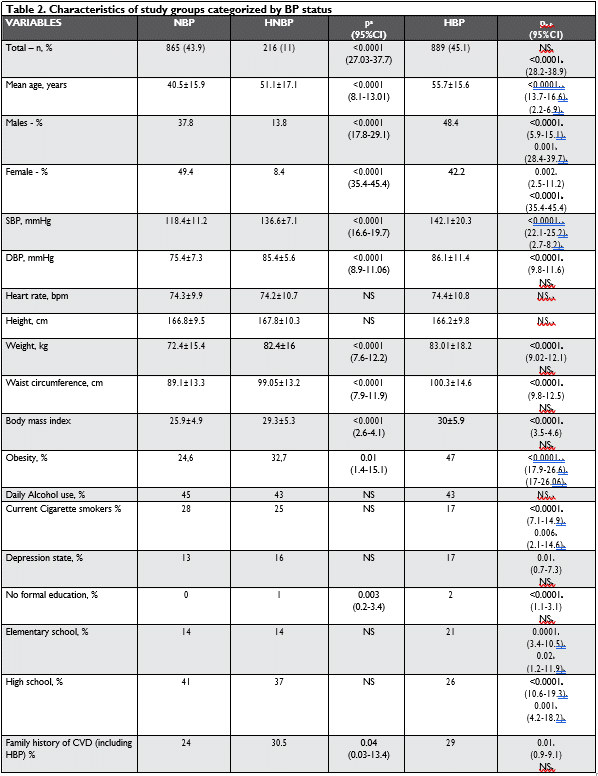
Table 2 shows the characteristics of the three categories of BP groups.
Values of systolic blood pressure (SBP), diastolic blood pressure (DBP), weight, waist circumference, BMI, TC, LDL and HDL cholesterol, TG, fasting blood glucose (FBG), glycated hemoglobin HbA1c, uric acid, serum creatinine, e GFR CKD – EPI and albumin/crea-tinine ratio were significantly highest in HNBP subjects than in NBP. There is no significant difference for the-se values between HNBP and HBP subjects.
NBP and HNBP subjects have the biggest number of cigarette smokers in the three groups and there were no differences in the consumption of alcohol.
The prevalence of „no formal” and elementary edu-cation increased steadily with the group who had in-creased BP, while the proportion of “high school” was significantly lower in HNBP and HBP groups.
As expected, salt intake is signifi cantly higher in HBP and HNBP subjects, compared with NBP (13.1 ± 4.1 vs 12.8 ± 3.6 vs 11.2±3.6 g/day, <0.0001), with no significant differences between HBP and HNBP in-dividuals.
Arterial stiffness and ankle brachial index (ABI) measurements showed that aortic pulse wave velocity (Ao PWV) was significantly higher in HNBP and HBP while an ABI < 0.9 was more frequent in HBP group.
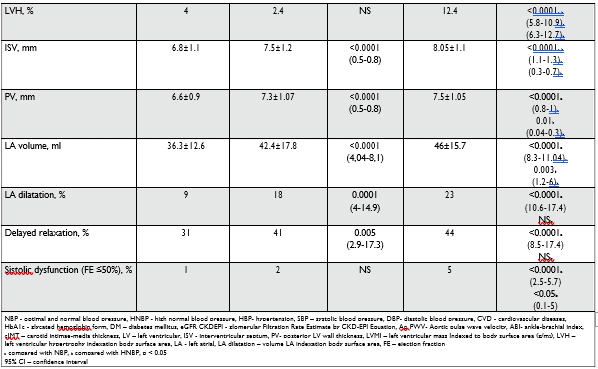
The evaluation of carotid arteries showed a higher intimae-media thickness (cIMT) in HNBP and HBP, with more frequent instable plaques in the HBP group. Transthoracic echocardiography (TTE) measure-ments showed values of left ventricular mass indexed to body surface area (LVMI), interventricular sep-tum (ISV) and posterior left ventricle wall thickness (PV), left atrial (LA) volume and LA dilatation, highest in HNBP and HBP groups than in NBP. The rates of delayed and impaired relaxation as signs of diastolic dysfunction, calculated by the study of E/A and E/e’ ratio was 44% in HBP vs 41% in HNBP (NS) vs 31% in NBP (p < 0.0001), being significantly more frequent in HBP and HNBP groups. Left ventricular hypertrophy indexation body surface area (LVH) and the systolic dysfunction of LV (efection fraction – FE ≤ 50%) also increases in parallel, with the BP values being more frequent in the HBP group.
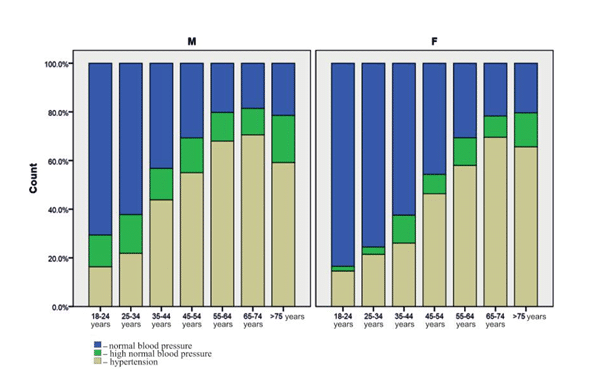
Figure 1. Prevalence of each BP status by gender and age.
- Association of risk factors with HNBP and
HBP
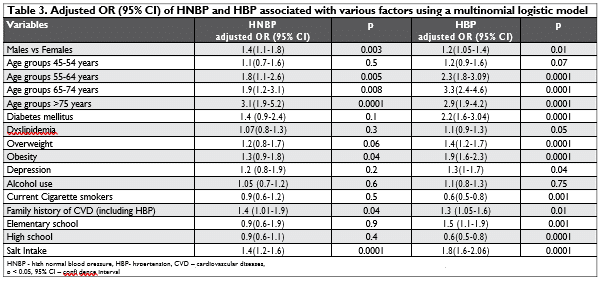
The multivariable-adjusted ORs of HNBP and HBP associated with various risk factors are presented in Table 3.
Males are more likely than females to have HNBP and to develop HBP. Age (beginning from 55 years) and family history of CVD (including HBP) are signifi-cantly associated with HNBP and HBP. DM and dysli-pidemia are risk factors for HNBP and significantly in-creased the risk of HBP. Overweight and obesity were risk factors for both HNBP and HBP. Depression as resulted by 13 items for evaluation of the state of de-pression was a risk factor for HBP, but in our study, it was not associated with HNBP. Compared to subjects with an elementary education status which predispo-sed them to HBP, those with a higher school educa-tion were less likely to have HNBP and HBP. Daily alcohol consumption (300 ml wine or 30 ml strong drinks) caused a modest but non-significant rise in the risk of HNBP and HBP. Cigarette smoking was not associated with HNBP and was also found to have a significantly negative association with HBP. Finally, salt intake is significantly associated with HNBP and HBP, regardless of age or sex.
After adjusting for age, gender, and family history of CVD/HBP, individuals with overweight/obesity and those with a high salt intake showed an increased risk for HNBP: OR 1.62, CI 1.32-1.98, p< 0.001, respec-tively OR 2.12, CI 1.67-2.68, p< 0.001. Clustering of these 2 factors was associated with a 3.52 higher OR (CI 2.78-4.76, p< 0.001) of HNBP compared with ab-sence of the association.
- Study groups related comorbidities and the risk of CVD
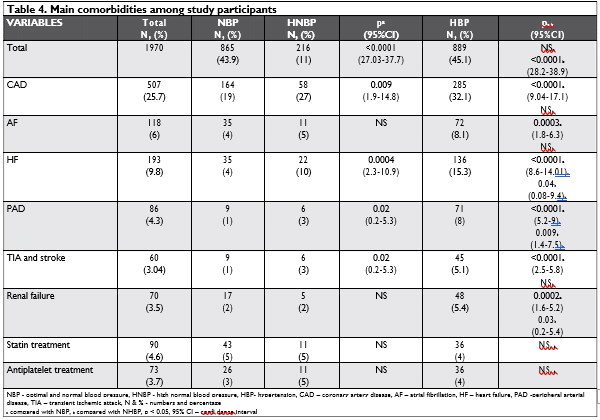
Related comorbidities among NBP, HNBP and HBP subjects are presented in Table 4.
Although the majority of the HBP subjects identi-fied in the SEPHAR III survey (78%) had at least one comorbidity, in the group of HNBP subjects there were 50% and only 30% of those with NBP. The rates of CHD, HF, PAD, TIA and stroke were significantly highest in HNBP and HBP patients compared with NBP, whereas the rates of AF and RF were correlated with the HBP status. Nevertheless, the use of statins and antiplatelet treatment was not frequent (from 3 to 5%) with no significantly differences between the groups.
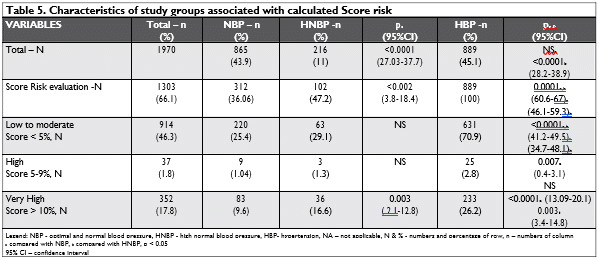
CV risk estimation using SCORE risk estimation was possible for 1303 (66.1 %) subjects: 312 (36.06 %) in NBP, 102 (47.2 %) in HNBP and 889 (100%) in HBP subjects. Table 5 shows the characteristics of study groups associated with calculated Score risk.
In each category of Score Risk, the number of HBP subjects is significantly greater than in NBP and HNBP subjects. Compared with the total number of subjects in each category of Score Risk, there are no differences in percentage of subjects with low to mo-derate risk between NBP and HNBP group, but are more with very high risk in HNBP than in NBP group; p=0.003, 95% CI: 2.1-12.8. Also, if we compare the percentage of subjects with high or very high risk in the group of HNBP vs NBP, there are more in HNBP group: 18.05 % (39 from 216) vs 10.6 % (92 from 865); p=0.002, 95% CI: 2.3-13.4
DISCUSSION
This study focused on a lot of adults aged between 18 and 80 years, representative of the Romanian po-pulation. The prevalence of HNBP was 11% (13.8% in males and 8.4% in females) and the prevalence of HBP was 45.1 % (48.4% in males and 42.2% in females) in all participants, which together means ~ 56% of popula-tion. By extrapolating the results from the SEPHAR III survey to the entire adult population of Romania, we can estimate that in 2016, there were approximately 7.4 million Romanian HBP patients and 1.8–1.9 million Romanian HNBP adult subjects. The latest represents a cohort associated with an increased risk of incident HBP at a rate of 8–20% over 4 years and also associa-ted with increased risk of CVD4-7,12.
The prevalence of HNBP in our study was only 11%, which is significantly less than the 31% observed in American adults and 32.8% observed in Netherlands12,13. Prevalence of HNBP was also less in Turkey, around 14.5% but is thought to be 36% in one 2011 meta-analysis, with a total sample of 250,741 individu-als14,15. The prevalence of HNBP in our sample is also considerably less compared to previously reported prevalence data in another European country, such as the 39.8% in Hungary16. In a report from the original cohorts of Brisighella Heart Study (Italy) and ENAH study (Croatia), HNBP prevalence was 25%, which is double than in our study17. However, our results are on the lower end of reported data for the prevalence of HNBP in other European regions (reported range of 30–40%)18. It’s well known that studies that exclu-ded individuals with HBP generally reported a higher prevalence of HNBP than those that included patients with HBP from the same countries, but this can’t ex-plicate the surprisingly lower prevalence of HBP in our sample4. The estimates of BP values in our study are somewhat different, involving the use of a special fully equipped medical caravan – the SEPHAR Bus, whereas other studies were based on selected population or only from one region of a country. By having a unique design (it allowed covering all 82 sites across the Ro-mania in a small period of time), a complete evaluati-on of all participants was possible, including rigorous BP measurements, minimizing the variation of BP and offering an estimation of a real trend in BP characteris-tics10,11. In addition, the prevalence of 45.1% HBP is on the higher end between European Countries, in con-trast with findings from recent epidemiological studies in Western Europe and could explain distribution analysis ascertained that the majority of participants have HBP19,20.
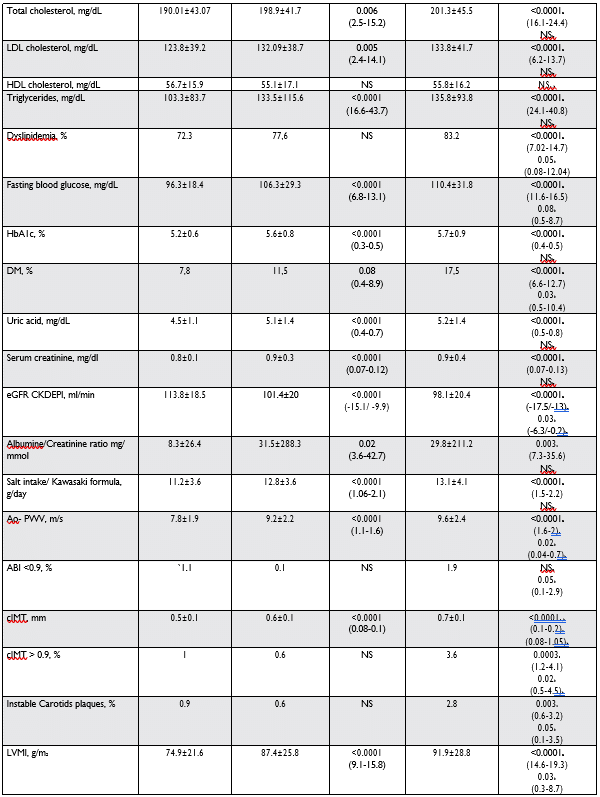
HNBP individuals appear to have a greater preva-lence of traditional CVD risk factors, compared to those with NBP. In this study, waist circumference, BMI, TC, LDL and HDL cholesterol, TG, FBG, HbA1c, uric acid, serum creatinine, e GFR CKD – EPI and al-bumine/creatinine ratio were signifi cantly highest in HNBP subjects than in NBP. There is no significant difference for these values between HNBP and HBP subjects and the results are generally concordant with the other studies5,12-21.
The multiple logistic regression analysis show-ed that male sex, age > 55 years, overweight, obe-sity, and salt intake, were signifi cantly associated with both HNBP and HBP. In addition, dyslipidemia, DM, depression state and a low level of education were significantly associated with HBP. However, a high education level was shown to be a protective factor, suggesting, as in other studies, that those with a hi-gher education were better informed about hyperten-sion and subsequently had a healthier lifestyle21. In our study, alcohol use was not a predictor of HNBP or HBP, and we also found that smoking appeared to be a protection factor for HBP [OR 0.60, CI 0.48-0.75, p= 0.001] (Table 3). Pooled analysis of ten smoking studies investigated the association between smoking and HNBP, and eight studies reported drinking status in 16,557 individuals with HNBP. All of these showed conflicting results, whereas some analyses suggested that individuals who smoke may lower BP compared with non-smoking individuals15,22,23. The relationship between smoking and development of HBP is still un-clear and controversial, but it was noted that a lower BP in smokers than non-smokers might be ascribed to the effect of smoking reducing weight24. Like in other studies, an index BMI, which defined overweight [OR 1.18, CI 0.82-1.70, p=0.06] and obesity [OR 1.33, CI 0.98-1.81, p=0.04], was a strong modifi able predictor of HNBP and HBP4,5,15,16 (Table 3).
SEPHAR III, based on the Kawasaki formula, estima-tes the salt intake for first time in a representative co-hort for the general adult population of Romania10,11. As expected, salt intake is significantly associated with HNBP and HBP regardless of age or sex, being signifi – cantly higher than in NBP (Table 3). There is no signi-ficant difference for these values between HNBP and HBP subjects and similar to other Central/East Euro-pean Countries, daily salt intake in Romania is almost double beyond the recommended intake by current guidelines3.
Arterial stiffness measurements estimated by Ao PWV were significantly higher in HNBP and HBP, whi-le an ABI <0.9 was more frequent in the HBP group. Our results confirm previous fi ndings that claim that arterial functions are impaired even at the prehyper-tensive stage25,26. As in other studies, the evaluation of carotid arteries showed a higher cIMT in HNBP and HBP, with more frequents instable plaques in HBP group27,28.
TTE measurements showed values of LVMI, ISV, PV, LA volume and LA dilatation, being highest in HNBP and HBP groups than in NBP. The MONICA/ KORA Augsburg trial was a study of individuals with HNBP with a follow-up of ten years, which found a significantly greater age-related increase in LV wall thickness (11.9 vs 4.7%, p<0.001) and LV mass (15.7 vs 8.6%, p=0.006) and an increased incidence of LV concentric remodelling (hazard ratio (HR) 10.7; 95% CI 2.82–40.4) and LVH (HR 5.3; 95% CI 1.58–17.9), compared with individuals with NBP29. The rates of delayed relaxation were more frequent in HBP and HNBP groups, whereas LVH and the systolic dysfunc-tion were more frequent in HBP subjects. Few studies have shown an association between the diastolic dys-function and HNBP status, but in our study, the rates of delayed relaxation, LA volume augmentation and LA dilatation like markers of diastolic dysfunction, ap-pears to be signifi cantly more frequent in HNBP than in NBP individuals, with no differences between HNBP and HBP. Our data confirm the continuous relations-hip between increasing degree of BP and deterioration of diastolic dysfunction, showing that changes in dias-tolic function are already present in prehypertensive stages30-33.
Similar to others studies, hypertension related co-morbidities are significantly higher in HBP and HNBP groups than in NBP individuals: 78% HBP participants identified in the SEPHAR III survey had at least one co-morbidity, 50% in the group of HNBP and 30% betwe-en those with NBP34-36. The rates of CHD, HF, PAD, TIA and stroke were significantly higher in HNBP and HBP patients compared with NBP, whereas the rates of AF and RF were not significantly associated with NHBP but with HBP status. A total of 507 participants (25.7% of the total population) had CHD; 19% in NBP, 27% in HNBP and 32.1% in HBP. This is extremely high for a population representative of the adult po-pulation aged 18-< 80 years, but in Romania although there are important limitations regarding the data-col-lection system, there is a clear tendency of increasing mortality due to ischemic heart disease. According to different international statistics, Romania holds fourth place in the world in terms of mortality due to ische-mic heart disease and stroke in men and third place in women7,37. In addition, a marker of subclinical disease, like Albu-mine/Creatinine ratio, is significantly higher in HNBP than in NBP individuals: 31.5 ± 288.3 vs 8.3 ± 26.4 mg/mmol, p=0.02. There is no significant difference in these values between HNBP and HBP subjects (31.5 3 vs 29.8 ± 211.2 mg/mmol) but it is evident that increases in Albumine/Creatinine ratio, parallel BP and antedate development of HBP38. Interestingly, and as suggested by 2018 ESC Hypertension Guidelines, in HNBP and HBP groups, we found an increase in serum uric acid to levels lower than those typically associated with gout but significantly higher than NBP individuals3 (Table 3).
The present study showed that 47.2 % of adults with HNBP had at least one of the following CVD risk factors (dyslipidemia, DM, overweight/obesity) and 18.05% were at high or very high cardiovascular risk, as estimated by the SCORE system. If we compare the percentage of subjects with high or very high risk in the group of HNBP vs NBP, there are more in HNBP group: 18.05 % vs 10.6 %; p=0.002, 95% CI: 2.3-13.4 (Table 5). Since HNBP is a phase in the progression to HBP, this might imply that almost half of individu-als with HNBP, and especially those at high and very high cardiovascular risk (almost 1 for 5), are at risk of hypertension and other CVDs4-7. By extrapolating the results from the SEPHAR III survey to the entire adult population of Romania, we can estimate that from 1.8–1.9 million Romanian individuals having HNBP in 2016, there are now at least 250000–300000 more HBP subjects to be added out of the estimated 7.4 million adult Romanian population at the time of sur-vey11,39.
LIMITATIONS AND STRENGTHS OF SEPHAR III SURVEY
SEPHAR III methodology enables a complete estima-tion of BP trends and a complete target organ dama-ge evaluation10,11. The strengths of the study include the large sample size associated with the principle of equality of chances of being enrolled in the study, re-gardless of the size of the place of residency and direct measurement of BP, rather than self-reported values. Use of the automated model OMRON M6 with an adjustable cuff for arm circumferences from 24 to 42 cm, respecting the current guideline recommendati-ons of the ESH/ESC provided a reliable measurement of BP and was beneficial in eliminating biases related to the traditional manual BP measurement2,3. The res-ponse rate in SEPHAR III survey was good (72.58%) but even that, the results of this cross-sectional study may not entirely reflect the health status of the gene-ral population in Romania, since the study population represented a convenience sample of those who sig-ned written consent to participate: 2124 respectively, with 1970 study participants with eligible data from the total number of 4226 randomly selected addresses from 84 study sites all around the country.
CONCLUSIONS
Individuals with HNBP represent ~11% of the popu-lation and had a higher proportion of cardiovascular risk factors when compared with normotensives. This might imply that they are at risk of HBP and others CVD. HNBP and HBP combined affl icts ~56% of Ro-manian adults (18–80 years). Possible explanations of this trend may be the following: unhealthy lifestyle and diet, including increased salt intake and the increase rate of obesity and DM. It’s of paramount importance to inform and educate the general public and health care providers not only about HBP but also to be awa-re of HNBP subjects at risk for cardiovascular diseases and of steps that should be taken to treat modifi able risk factors in these people.
Conflict of interest: none declared.
Acknowledgements: The authors wish to express their gratitude to the team of the SEPHAR study (http://www.sephar.ro/) for the data used in this article.
References
1. Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Den-nison Himmelfarb C, DePalma SM, Gidding S, Jamerson KA, Jones DW, MacLaughlin EJ, Muntner P, Ovbiagele B, Smith SC Jr, Spen-cer CC, Stafford RS, Taler SJ, Thomas RJ, Williams KA Sr, William-son JD, Wright JT. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Re-port of the American College of Cardiology/American Heart Asso-ciation Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2017; DOI: 10.1016/j.jacc.2017.11.005
2. Mancia G, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Böhm M, Christiaens T, Cifkova R, De Backer G, Dominiczak A, Galderisi M, Grobbee DE, Jaarsma T, Kirchhof P, Kjeldsen SE, Laurent S, Manolis AJ, Nilsson PM, Ruilope LM, Schmieder RE, Sirnes PA, Sleight P, Viigi-maa M, Waeber B, Zannad F, Redon J, Dominiczak A, Narkiewicz K, Nilsson PM, Burnier M, Viigimaa M, Ambrosioni E, Caufield M, Coca A, Olsen MH, Schmieder RE, Tsioufis C, van de Borne P, Zamorano JL, Achenbach S, Baumgartner H, Bax JJ, Bueno H, Dean V, Deaton C, Erol C, Fagard R, Ferrari R, Hasdai D, Hoes AW, Kirchhof P, Kn-uuti J, Kolh P, Lancellotti P, Linhart A, Nihoyannopoulos P, Piepoli MF, Ponikowski P, Sirnes PA, Tamargo JL, Tendera M, Torbicki A, Wijns W, Windecker S, Clement DL, Coca A, Gillebert TC, Ten-dera M, Rosei EA, Ambrosioni E, Anker SD, Bauersachs J, Hitij JB, Caulfield M, De Buyzere M, De Geest S, Derumeaux GA, Erdine S, Farsang C, Funck-Brentano C, Gerc V, Germano G, Gielen S, Haller H, Hoes AW, Jordan J, Kahan T, Komajda M, Lovic D, Mahrholdt H, Olsen MH, Ostergren J, Parati G, Perk J, Polonia J, Popescu BA, Reiner Z, Rydén L, Sirenko Y, Stanton A, Struijker-Boudier H, Tsiou-fi s C, van de Borne P, Vlachopoulos C, Volpe M, Wood DA. 2013 ESH-ESC Guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Soci-ety of Cardiology. J Hypertens. 2013; 31:1281–1357.
3. Williams B; Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burni-er M, Clement DL, Coca A, de Simone G, Dominiczak A, Kahan T, Mahfoud F, Redon J, Ruilope L, Zanchetti A, Kerins M, Kjeldsen SE, Kreutz R, Laurent S, Lip GYH, McManus R, Narkiewicz K, Ruschitz-ka F, Schmieder RE, Shlyakhto E, Tsioufis C, Aboyans V, Desormais I. 2018 ESC/ESH Guidelines for the management of arterial hyperten-sion.The Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Soci-ety of Hypertension (ESH). European Heart Journal. 2018; 39:3021– 3104.
4. Egan BM, Stevens-Fabry S. Prehypertension — prevalence, health risks, and management strategies. Nat. Rev. Cardiol.2015;12, 289– 300.
5. Vasan RS, Larson MG, Leip EP, Kannel WB, Levy D. Assessment of frequency of progression to hypertension in non-hypertensive par-ticipants in the Framingham Heart Study: a cohort study. Lancet. 2001; 358: 1682–1686.
6. Vasan RS, Larson MG, Leip EP, Evans JC, O›Donnell CJ, Kannel WB, Levy D: Impact of high-normal blood pressure on the risk of cardio-vascular disease. N Engl J Med. 2001; 345: 1291–1297.
7. Piepoli MF, Hoes AW, Agewall S, Albus C, Brotons C, Catapano AL, Cooney MT, Corrà U, Cosyns B, Deaton C, Graham I, Hall MS, Hobbs FDR, Løchen ML, Löllgen H, Marques-Vidal P, Perk J, Prescott E, Redon J, Richter DJ, Sattar N, Smulders Y, Tiberi M, van der Worp HB, van Dis I, Verschuren WMM, Binno S. 2016 European Guide-lines on cardiovascular disease prevention in clinical practice. Euro-pean Heart Journal. 2016;37:2315–2381.
8. Dorobanţu M, Darabont R, Ghiorghe S, Arsenescu-Georgescu C, Macarie C, Mitu F, Lighezan D, Musetescu R, Pop C, Ardeleanu E, Craiu E, Tăutu OF. Hypertension prevalence and control in Romania at a seven-year interval. Comparison of SEPHAR I and II surveys. J Hypertens. 2014; 32:39–47.
9. Dorobantu M, Darabont RO, Badila E, Ghiorghe S. Prevalence, awareness, treatment, and control of hypertension in Romania: re-sults of the SEPHAR study. International Journal of Hypertension. 2010; 2010: 970694, https://doi.org/10.4061/2010/970694.
10. Dorobantu M, Darabont R, Dimulescu D, Sinescu C, Gusbeth PT, Georgescu CA, Mitu F, Lighezan D, Pop C, Babes K, Giuca A, Brinza I, Udrescu M, Herdea V, Tautu O. New national epidemiological sur-vey for the assessment of trend in hypertension’s prevalence, treat-ment and control among the adult population of Romania: SEPHAR III: design and methodology. J Hypertens Res. 2016; 2:143–152.
11. Dorobantu M, Tautu OF, Dimulescu D, Sinescu C, Gusbeth-Tatomir P, Arsenescu-Georgescu C, Mitu F, Lighezan D, Pop C, Babes K, Gi-uca A, Branza I, Udrescu M, Herdea V, Darabont R. Perspectives on hypertension’s prevalence, treatment and control in a high cardio-vascular risk East European country: data from the SEPHAR III sur-vey. J Hypertens. 2018;36(3):690 –700.
12. Wang Y, Wang QJ: The prevalence of prehypertension and hyper-tension among US adults according to the new joint national com-mittee guidelines: new challenges of the old problem. Arch Intern Med. 2004;164: 2126–2134.
13. Agyemang C, van Valkengoed I, van den Born BJ, Stronks K. Prev-alence and determinants of prehypertension among African Suri-namese, Hindustani Surinamese, and White Dutch in Amsterdam, the Netherlands: the SUNSET study. Eur J Cardiovasc Prev Rehabil. 2007;14(6):775-781.
14. Erem C, Hacihasanoglu A, Kocak M, Deger O, Topbas M. Preva-lence of prehypertension and hypertension and associated risk fac-tors among Turkish adults: Trabzon Hypertension Study. J Public Health. 2009;31(1):47-58.
15. Guo X, Zou L, Zhang X, Guo X, Zou L, Zhang X, Li J, Zheng L, Sun Z, Hu J, Wong ND, SuN. Prehypertension: A Meta-Analysis of the Epidemiology, Risk Factors, and Predictors of Progression. Texas Heart Institute Journal. 2011;38(6):643-652.
16. Sonkodi B, Sonkodi S, Steiner S, Helis E, Turton P, Zachar P, Abrahám G, Legrady P, Fodor JG. High Prevalence of Prehyperten-sion and Hypertension in a Working Population in Hungary. Ameri-can Journal of Hypertension. 2012; 2:204-208.
17. Ivkovic V, Parini A, Vrdoljak A, Sertic J, Laganovic M, Arrigo FGC. [PP.19.05] Prehypertension in european rural population – data from the Brisghella heart study (Italy) and Enah study (Croatia). Journal of Hypertension. 2016; 34: e234.
18. Costanzo S, Di Castelnuovo A, Zito F, Krogh V, Siani A, Arnout J, Cappuccio FP, Miller MA, van Dongen M, de Lorgeril M, de Gaetano G, Donati MB, Iacoviello L. European Collaborative Group of the IMMIDIET project. Prevalence, awareness, treatment and control of hypertension in healthy unrelated male-female pairs of European re-gions: the dietary habit profile in European communities with differ-ent risk of myocardial infarction–the impact of migration as a mod-el of gene-environment interaction project. Journal of Hypertens. 2008; 26:2303–2311.
19. Bielecka-Dabrowa A, Aronow WS, Rysz J, Banach M. The rise and fall of hypertension: lessons learned from Eastern Europe. Curr Car-diovasc Risk Rep. 2011; 5:174–179.
20. Danaei G, Finucane MM, Lin JK, Singh GM, Paciorek CJ, Cowan MJ, Farzadfar F, Stevens GA, Lim SS, Riley LM, Ezzati M. Global Burden of Metabolic Risk Factors of Chronic Diseases Collaborating Group (Blood Pressure). National, regional, and global trends in systolic blood pressure since 1980: systematic analysis of health examination surveys and epidemiological studies with 786 country-years and 5.4 million participants. Lancet. 2011; 377:568–577.
21. Lin Y, Lai X, Chen G, Xu Y, Huang B, Chen Z, Zhu S, Yao J, Jiang Q, Huang H. Prevalence and Risk Factors Associated with Prehyperten-sion and Hypertension in the Chinese She Population. Kidney Blood Press Res. 2012; 35:305–313.
22. Okubo Y, Miyamoto T, Suwazono Y, Kobayashi E, Nogawa K. An as-sociation between smoking habits and blood pressure in normoten-sive Japanese men. J Hum Hypertens. 2002; 16: 91–96.
23. Okubo Y, Suwazono Y, Kobayashi E, Kobayashi E, Nogawa K. An as-sociation between smoking habits and blood pressure in normoten-sive Japanese men: a 5-year follow-up study. Drug Alcohol Depend. 2004; 73: 167–174.
24. Primatesta P, Falaschetti E, Gupta S, Marmot MG, Poulter NR. As-sociation between smoking and blood pressure: evidence from the health survey for England. Hypertension. 2001; 37: 187–193.
25. Gedikli O, Kiris A, Ozturk S, Baltaci D, Karaman K, Durmus I, Baykan M, Celik S. Effects of Prehypertension on Arterial Stiffness and Wave Reflections. Clinical and Experimental Hypertension. 2010; 32(2):84-89.
26. Tomiyama H, Yamashina A. Arterial Stiffness in Prehypertension: A Possible Vicious Cycle. Journal of Cardiovascular Translational Re-search. 2012; 5 (3): 280-286.
27. Manios E, Tsivgoulis G, Koroboki EA, Stamatelopoulos K, Papami-chael C. Impact of prehypertension on common carotid artery in-tima-media thickness and left ventricular mass. Stroke. 2009; 40: 1515-1518.
28. Kim SH, Cho GY, Baik I, Lim SY, Choi CU, Lim HE, Kim EJ, Park CG, Park J, Kim J, Shin C. Early Abnormalities of Cardiovascular Struc-ture and Function in Middle-Aged Korean Adults with Prehyperten-sion: The Korean Genome Epidemiology Study, American Journal of Hypertension. 2011; 2 (1): 218–224.
29. Markus MR, Stritzke J, Lieb W, Mayer B, Luchner A, Döring A, Keil U, Hense HW, Schunkert H. Implications of persistent prehyperten-sion for ageing related changes in left ventricular geometry and func-tion: the MONICA/KORA Augsburg study. Journal of Hypertens. 2008; 26:2040–2049.
30. Yeter E, Akçay M, Keleş T, Durmaz T, Bayram NA, Ozdemir L. The association of diastolic dysfunction and circadian variation of blood pressure in prehypertension. J Am Soc Echocardiogr. 2009 ;22(6):726-731.
31. Jang SY, Kim S, Lee CK, Cho EJ, Cho SJ, Lee SC.Prehypertension and Left Ventricular Diastolic Dysfunction in Middle-Aged Koreans. Ko-rean Circulation Journal. 2016;46(4):536-541.
32. Lopes RL, Carvalho RF, Vilela EM, Bettencourt P, Moreira A, Aze-vedo A. Diastolic Function Is Impaired in Patients with Prehypertension: Data from the EPI Porto Study. Rev Esp Cardiol. 2017.http:// dx.doi.org/10.1016/j.rec.2017.11.015
33. Celik T, Yuksel UC, Fici F, Celik M, Yaman H, Kilic S, Iyisoy A, Dell’oro R, Grassi G, Yokusoglu M, Mancia G. Vascular inflammation and aortic stiffness relate to early left ventricular diastolic dysfunc-tion in prehypertension, Blood Pressure. 2013; 22(2): 94-100.
34. Vasan RS, Larson MG, Leip EP, Evans JC, O’Donnell CJ, Kannel WB, Levy D. Impact of high-normal blood pressure on the risk of cardio-vascular disease. N. Engl. J. Med. 2001; 345:1291-1297.
35. Hsia J, Margolis KL, Eaton CB, Wenger NK, Allison M, Wu L, LaC-roix AZ, Black HR. Prehypertension and cardiovascular disease risk in the Women’s Health Initiative. Circulation. 2007; 115: 855-860.
36. Mainous AG. 3rd, Everett CJ, Liszka H, King DE, Egan BM. Prehy-pertension and mortality in a nationally representative cohort. Am. J. Cardiol. 2004; 94: 1496-1500.
37. Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB, Bravata DM, Dai S, Ford ES, Fox CS, Franco S, Fullerton HJ, Gillespie C, Hailpern SM, Heit JA, Howard VJ, Huffman MD, Kis-sela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Magid D, Marcus GM, Marelli A, Matchar DB, McGuire DK, Mohler ER, Moy CS, Mussolino ME, Nichol G, Paynter NP, Schreiner PJ, Sorlie PD, Stein J, Turan TN, Virani SS, Wong ND, Woo D, Turner MB;. Heart Disease and Stroke Statistics – 2013 update: a report from the American Heart Association. Circulation. 2013;127(1): e6–e245.
38. Ninomiya T, Kubo M, Doi Y, Yonemoto K, Tanizaki Y, Tsuruya K, Sueishi K, Tsuneyoshi M, Iida M, Kiyohara Y. Prehypertension in-creases the risk for renal arteriosclerosis in autopsies: the Hisayama study. J Am Soc Nephrol. 2007; 18:2135–2142.
39. Geldsetzer P, Manne-Goehler J, Marcus ME, Ebert C, Zhumadilov Z, Wesseh CS, Tsabedze L, Supiyev A, Sturua L, Bahendeka SK, Sibai AM, Quesnel-Crooks S, Norov B, Mwangi KJ, Mwalim O, Wong-McClure R, Mayige MT, Martins JS, Lunet N, Labadarios D, Karki KB, Kagaruki GB, Jorgensen JMA, Hwalla NC, Houinato D, Houehanou C, Msaidié M, Guwatudde D, Gurung MS, Gathecha C, Doroban-tu M, Damasceno A, Bovet P, Bicaba BW, Aryal KK, Andall-Brere-ton G, Agoudavi K, Stokes A, Davies JI, Bärnighausen T, Atun R, Vollmer S, Jaacks LM. The state of hypertension care in 44 low-income and middle-income countries: a cross-sectional study of na-tionally representative individual-level data from 1·1 million adults. Lancet.2019:https://doi.org/10.1016/S0140-6736(19)30955-9.
 This work is licensed under a
This work is licensed under a