Download PDF
https://doi.org/10.47803/rjc.2020.30.3.437
Radu Brezeanu1, Adrian Bucsa2, Gabriel Stanica3, Carmen Beladan2, Carmen Ginghina2, Dan Deleanu2, Lucian Predescu2
1 „Bagdasar-Arseni” Clinical Emergency Hospital, Bucharest, Romania
2 Department of Cardiology, „Prof. CC Iliescu” Institute for Cardiovascular Diseases, Bucharest, Romania
3 Pitesti Clinical Emergency Hospital, Romania
Abstract: The recurrence of ischemic events in patients with prior coronary artery bypass graft surgery (CABG) is an actual and challenging problem. Advances in surgical techniques and intensive care treatment have decreased the short-term mortality after CABG. Nevertheless, the increase in age and comorbidities of patients referred for CABG impacts the likelihood of graft degeneration or graft failure. More patients are referred to the cardiologist after CABG for the recurrence of symptoms. Particularities in the diagnosis and treatment of these patients need to be considered by the interventional cardiologist to ensure good angiographic and clinical results.
Keywords: bypass, graft, failure.
Rezumat: Recurenţa evenimentelor ischemice la pacienţii ce au beneficiat de revascularizare miocardică chirurgicală prin by-pass aorto-coronarian (BAC) este o problemă dificilă şi de actualitate. Avansul tehnicilor chirurgicale şi a măsurilor de terapie intensivă postoperatorie au scăzut mortalitatea pe termen scurt post BAC. Cu toate acestea creşterea vârstei şi a comorbidităţilor pacienţilor influenţează morbiditatea pe termen mediu şi lung prin degenerarea grafturilor. Un număr mai mare de pacienţi sunt îndrumaţi către cardiolog pentru reapariţia simptomelor după BAC. Particularităţile de diagnostic şi tratament ale acestor pacienţi trebuie să fie cunoscute cardiologului intervenţionist pentru a asigura un rezultat angiografic şi clinic favorabil.
Cuvinte cheie: bypass, graft, degenerare.
INTRODUCTION
Performing percutaneous coronary intervention (PCI) in patients who previously had coronary artery bypass surgery (CABG) is often required to relieve angina due to graft failure. PCI after prior CABG is more difficult to perform, technically complex and associated with increased procedural risk. However, despite all this limitations, PCI is the preferred method for secondary revascularization in patients who had previously undergone CABG. Understanding the particularities of graft failure and the expected results of angioplasty and stenting of the graft are important to decide on the best modality of revascularization. Additional diagnostic techniques such as intracoronary imaging and adjuvant PCI techniques can improve the final result.
CASE PRESENTATION
We present the case of a 76-year-old woman, who presented to our hospital with unstable angina. Her past medical history included CABG with left internal mammary artery (LIMA) on the left anterior descending artery (LAD) and saphenous venous graft (SVG) on first obtuse marginal (OM) for severe left main coronary artery stenosis (one year ago). Her cardiovascular risk factors were stage III hypertension, dyslipidemia, and type II diabetes mellitus. On admission, her electrocardiogram showed sinus rhythm, heart rate of 72 bpm, ST-segment depression of 1 mm in the precordial leads. Echocardiography revealed a preserved ejection fraction and mild mitral regurgitation.
Coronary angiogram was performed and show-ed the following: severe calcification of left main and LAD, tight stenosis of left main and proximal LAD, occlusion of left circumflex artery (LCX), 50% stenosis of the right coronary artery at the ostium, severe ostial stenosis of SVG on first OM and occlusion of LIMA (Figures 1,2,3).
The patient was on maximal treatment for angina with high dose of beta-blocker, angiotensin-converting enzyme inhibitor, statin, long-acting nitrate and dual antiplatelet therapy.
Taking into account the risk of a redo-CABG (euroSCORE II was 6.78%), the difficulties to achie-ve a complete revascularization by surgery and the patient’s desire, the local heart team decided to revascularize the lesions by PCI. First, we addressed the left coronary artery lesions. We used a 7 French 4.0 extra backup guiding catheter to engage the left coronary artery. Rotablation was done using a 1.5 mm burr, followed by pre-dilatation with a 2.5×20 mm non-compliant balloon at high pressure, with good plaque modifi cation and preparation (Figure 4).
Two drug eluting stents (DES) were implanted from LAD to the ostium of left main – a 3.0×24 mm DES followed by a 3.5×28 mm DES. The stents were post-dilated with a 3.5×15 mm noncompliant balloon, achieving a good angiographic result (Figure 5). After the procedure the patient stabilized, and she was dis-charged free from angina.
In another session, we addressed the lesion at the ostium of the SVG to first OM. We used a 6 French 4.0 Judkins right guiding catheter to engage the SVG. The lesion was predilated with a 3.0 mm non-com-pliant balloon. A 3.0×18 mm DES was implanted with good initial expansion. However, severe stent recoil was observed during contrast injection (Figure 6).
A non-compliant balloon was then used for post-dilatation, which expanded the stent completely, but subsequent contrast infusions after balloon deflation showed the same severe recoil.
To better understand the mechanism of stent re-coil we decided to perform intravascular ultrasound imaging (IVUS). IVUS showed important stent deformation with stent struts protruding into the vessel lumen, which raised the suspicion of stent fracture (Figure 7).
We decided to implant a 3.0x12mm DES in the pre-existing stent (Figure 8). IVUS showed good expansion of the stent and correction of initial stent deformation (Figure 9).
The patient underwent a good and uncomplicated recovery after the procedure and was discharged two days after PCI. Her recommended treatment was: dual antiplatelet therapy, beta-blocker, high dose statin, ACE- inhibitor and nitrate. She remains asymptomatic at 1 year follow up.
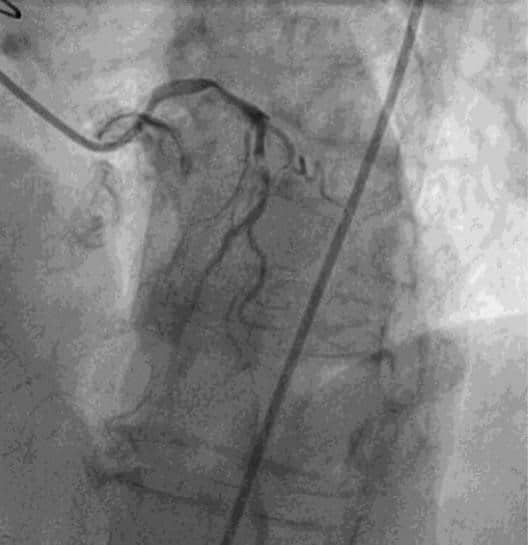
Figure 1. Coronary angiogram (left anterior oblique 300 cranial 300) – tight stenosis of left main and proximal left anterior descending artery, occlusion of left circumflex artery.
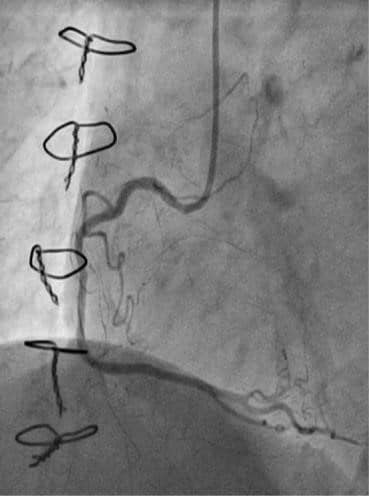
Figure 2. Coronary angiogram (left anterior oblique 300) – 50% stenosis of the right coronary artery at the ostium.
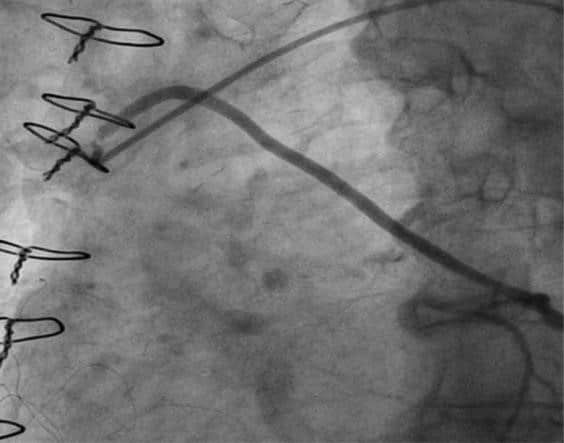
Figure 3. Coronary angiogram (left anterior oblique 300) – severe ostial stenosis of saphenous vein graft on first obtuse marginal.
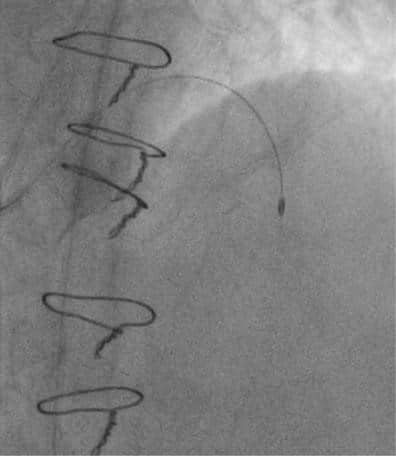
Figure 4. Rotablation of left main and left anterior descending artery using 1.5 mm burr.
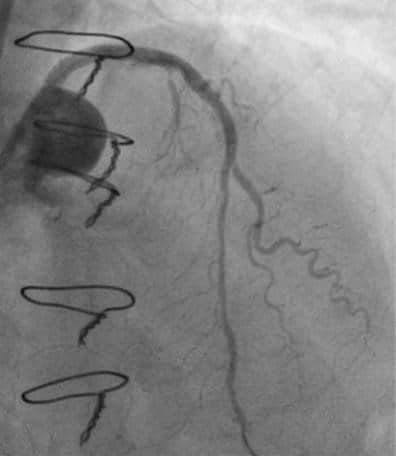
Figure 5. Angiographic result stenting of left main and left anterior descending artery.
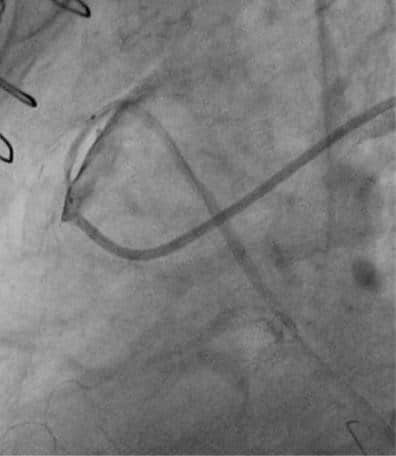
Figure 6. Severe stent recoil on saphenous vein graft on first obtuse marginal branch.
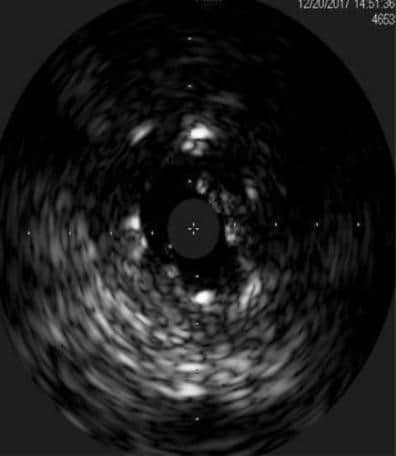
Figure 7. Intravascular ultrasound – suspected stent fracture on saphenous vein graft on first obtuse marginal branch.
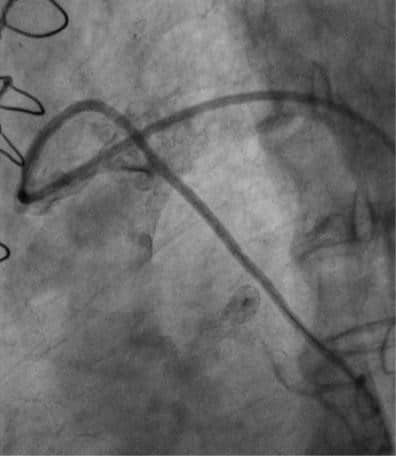
Figure 8. Angiographic result after second stent implantation on saphenous vein graft to first obtuse marginal branch.
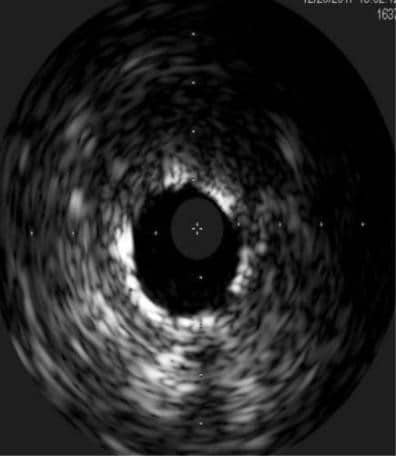
Figure 9. Intravascular ultrasound after second stent implantation on saphenous vein graft on first marginal branch.
DISCUSSIONS AND CLINICAL PRACTICE IMPLICATIONS
CABG or saphenous vein bypass graft disease (SVGD) is a challenging problem after surgical myocardial revascularization. The SVG remains the main graft mate-rial at present with 93% patency at 1-year post-CABG, 74% at 5 years and 41% at 10 years1. Other studies suggest that up to 12% of SVG become occluded at 6 months after CABG, with almost 3.5% occluding in the first month2. The SVGD is defined as a >50% stenosis of the graft associated with the recurrence of ischemic symptoms. Studies showed that the patency of grafts is better for arterial (85-95% patency for LIMA) compared with saphenous venous grafts (SVG patency 40%) and the annual rate of revascularization ranges from 8.6 to 10.4% after CABG. Blood stagnation, lipid accumulation, and thrombosis are more likely to occur in the SVG as the veins do not possess the thick elastic fibers and smooth muscle layers that are present in the arterial wall3.
Several risk factors have been identified for SVG failure such as surgery-related factors, impact of diabetes mellitus and endothelial dysfunction, dyslipidemia and smoking, low 25hydroxivitamin D level, high level of erythropoietin and other rheologic factors. The mechanism of SVG failure may be different with respect to the time from CABG. The thrombotic mechanism is associated with early graft occlusion (<1 month after CABG), abnormal hyperplasia of the neointima in the subacute phase (1-12 months), and atherosclerotic lesions in late SVG failure (>1 year after CABG)4. In our case we might suspect rapid progression of atherosclerosis in a patient with multiple cardiovascular risk-factors and possibly surgery related factors if we consider the premature closure of LIMA graft).
Considering all these factors the prevention and treatment of SVGD remains a clinical challenge. Graft stenosis can be treated with repeat surgical revascularization (redo-CABG) or PCI of the graft or on the native vessel. The redo-CABG is more often indicated in the younger patient without major comorbidities and who can benefit from an arterial graft (LIMA has not been used in prior CABG). If the LIMA has been used or the surgical risk is remarkably high a percutaneous revascularization strategy is recommended. The optimal treatment strategy in patients with graft failure should be decided by heart team, that involves consultation between the interventional cardiologist and the cardiovascular surgeon5.
Questions remain about the target of the PCI, with mainly two options: to treat the graft or the native vessels, both strategies having multiple challenges. When the graft is occluded the single feasible option is to performed PCI on the native arteries, because of the poor results following graft recanalization6. Our case highlights several aspects of each strategy and is-sues related to revascularization.
In the case of SVG PCI there are a couple of aspects that we want to mention:
1. Finding the grafts and engaging the guiding catheter may be challenging and this can lead to poor guide catheter support and difficulties in the delivery of the equipment (especially for distal stenosis). Choosing the appropriate guide catheter (such as multi-purpose for right coronary SVG, left amplatz, or left coronary bypass for left coronary SVG) or guide catheter extension (Guideliner, Guidezilla) can improve support during PCI7.
2. PCI of SVG have a higher risk for distal embolization and periprocedural myocardial infarction. This can be reduced by using an embolic protection device (EPD) such as Guardwire (Medtronic Vascular) – distal wire mounted balloon, or Filterwire (Boston Scientific) and Spider (EV3)
– both filters. EPD is not indicated for in-stent restenosis (fibrous tissue with low risk of emboli-zation), SVG that is less than 2 years old (no time to develop degeneration predisposing to embolization) and distal or anastomotic lesions8.
3. Adjunctive pharmacotherapy such as vasodila-tors (adenosine, nitroglycerine, nicardipine, vera-pamil) can be used before and after PCI if slow flow occurs. Glycoprotein IIB/IIIA inhibitors are reserved for cases with large thrombus burden9.
4. Intravascular imaging (IVUS, optic coherence to-mography) can be used to determine the optimal stent dimension and to evaluate the result after PCI. In our case, IVUS was performed after PCI of the ostium of SVG that demonstrated under expansion and stent fracture. Not recognizing such a situation can lead to stent thrombosis or restenosis and impact the rate of major adverse cardiac events10,11
If the native arteries are the target for the PCI, we need to consider other challenges:
- Complex lesions can require special PCI techniques such as calcified lesions that often need debulking methods such as rotational atherectomy or more recently intravascular balloon lithotripsy (Shockwave Medical)12. However, using of rotational atherectomy involve a higher rate of PCI complication such as: vascular access complications, myocardial infarction, stroke, urgent CABG, and death, as well as angiographic complications, including dissection, short-term closure, perforation, side branch loss, and the slow-flow or no-reflow phenomenon.
In our case, we performed PCI of the left main for a heavily calcified lesion. The proper preparation using rotational atherectomy and good predilatation of the plaque ensures a good angiographic result.
- Incomplete revascularization can be the case in patients with chronic total occlusions (CTO) with no options for PCI. In our case, the left circumflex artery had a proximal CTO, but by sten-ting the SVG to the fi rst OM we ensure good coronary perfusion of the left circumflex artery territory.
In cases with poor left ventricular function, we may need a hemodynamic support during such high-risk PCI of the left main or when performing PCI on the last remaining vessel13. This is-sue can be overcome with the use of hemodynamic support systems such as intra-aortic balloon pump (IABP) or Impella system, which increases coronary perfusion and cardiac output during high-risk PCI. During this case we encounter a quite rare complication of PCI that worth our attention, stent fracture (SF)/deformation. Coronary SF was first reported in 2002 in a PCI with a BMS on a venous bypass graft, and for DES the first case was reported in 2004. The incidence of SF varies between 1 to 8% depending on the percentage of patient that undergo follow up and the imaging modality that is used. The degree of deformation (partial types to complete fracture) can also influence the clinical manifestation from asymptomatic in case of minor fracture to dramatic clinical manifestation such as sudden cardiac death in case of stent thrombosis14.
The predictors of SF can be divided into:
- Technical factors: balloon or stent overexpansion, stent overlap and lenght (higher radial force), stenting technique (the crush technique can increase the risk)
- Stent type and conformability: the degree to which a stent bend in longitudinal axis after implantation determine the response force from the vessel wall that tend to revert the axis of the wall. Sirolimus eluting stent (SES) were observed to have higher risk for SF and one possible explanation is the closed-cell structure of these stents that increase the rigidity15.
- Anatomic and target vessel characteristics: tortuous and angulated vessel, complex lesion (the length of the disease segment, degree of calcification), repetitive cardiac contraction that increase the compression, bending and shear stress on the stent struts. From several studies we know that SF is more common in right coronary artery and saphenous graft location, while left main stents are less likely to fracture16.
The diagnostic modalities influence the rate of detection of SF. Because stent visibility is limited on conventional fluoroscopy, additional imaging modalities such as stent boost, IVUS, OCT can be use either to confirm SF suggested by angiography or to detect deformation that were missed by angiography. IVUS has the advantage to provide additional information regarding stent failure such as vessel remodeling, peri-vascular tissue, stent expansion and malposition17. The management of SF depends on the type of fracture, presence of ischemia or factors that can predict recurrence. Option for treatment include prolonged dual antiplatelet treatment, balloon angioplasty or stenting the lesion and CABG in case we identify factor of recurrence (e.g. myocardial bridging, extrinsic compression)18. In our case we can speculate that SF was mainly ca-use by the increase rigidity of the aortic wall, that cause increase in the radial force and collapse of the stent struts. After we implanted the second stent there was no evidence of stent malposition on IVUS. We can also comment on the need to treat the 50% ostial lesion on the right coronary artery. We did not consider PCI in this session because of the complex and prolonged procedure on the LAD and SVG. The plan was to follow up the patient and to perform PCI in case of recurrence of symptoms. At one year follow up, with optimal medical treatment (including DAPT) patient had no angina and we decided to continue with prolonged DAPT. Considering all this aspect, percutaneous coronary interventions for graft failure or significant graft disease remains a challenge. The use of a complex PCI technique is usually necessary. PCI on degenerated SVG presents higher complication rate and worse clinical outcome compared with native arteries interventions. Rotational atherectomy may be especially useful at this type of patients for plaque modification and preparation to ensure a good expansion of the stents in severe calcified lesions. Intracoronary imaging plays an important role in planning and ensuring a good angiographic result, also. IVUS can better evaluate SAVG plaque, calcifications of native vessel, stent malfunctions, under-expansion or deformation. The operator experience and creating tertiary centers for this type of procedure can improve the rate of success and limit the complications.
Case highlights:
Complex PCI in a patient with severe grafts failure
The utility of rotational atherectomy in plaque modification and preparation to ensure a good expansion of the stents in severely calcified lesions
IVUS in a saphenous vein graft
The utility of IVUS in highlighting:
-stent malfunctions;
-the achievement of a good expansion of the stent;
-the correction of the stent under expansion and deformation.
Conflict of interest: none declared.
References
1. Goldman S, Zadina K, Moritz T, et al. Long-term patency of saphe-nous vein and left internal mammary artery grafts after coronary artery bypass surgery: results from a Department of Veterans Affairs Cooperative Study. J Am Coll Cardiol. 2004;44(11):2149-2156.
2. Fitzgibbon GM, Kafka HP, Leach AJ, Keon WJ, Hooper GD, Burton JR. Coronary bypass graft fate and patient outcome: angiographic follow-up of 5,065 grafts related to survival and reoperation in 1,388 patients during 25 years. J Am Coll Cardiol. 1996;28(3):616-626.
3. Motwani JG, Topol EJ. Aortocoronary saphenous vein graft disease: pathogenesis, predisposition, and prevention. Circulation. 1998; 97(9):916-931.
4. Lee MS, Park SJ, Kandzari DE, et al. Saphenous vein graft intervention. JACC Cardiovasc Interv. 2011;4(8):831-843.
5. Sabik JF 3rd, Raza S, Blackstone EH, Houghtaling PL, Lytle BW. Value of internal thoracic artery grafting to the left anterior descending coronary artery at coronary reoperation. J Am Coll Cardiol 2013; 61(3):302–310.
6. Levine, GN, Bates, ER, Blankenship, JC. “2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention. A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines and the Society for Cardio-vascular Angiography and Interventions”. J Am Coll Cardiol. vol. 58. 2011. pp. e44-122.
7. Brilakis, ES, Rao, SV, Banerjee, S. “Percutaneous coronary interven-tion in native arteries versus bypass grafts in prior coronary artery bypass grafting patients: a report from the national cardiovascular data registry”. JACC Cardiovasc Interv. vol. 4. 2011. pp. 844-50.
8. Baim, DS, Wahr, D, George, B. “Randomized trial of a distal embolic protection device during percutaneous intervention of saphenous vein aorto-coronary bypass grafts”. Circulation. vol. 105. 2002. pp. 1285-90.
9. Roffi, M, Mukherjee, D, Chew, DP. “Lack of benefit from intrave-nous platelet glycoprotein IIb/IIIa receptor inhibition as adjunctive treatment for percutaneous interventions of aortocoronary bypass grafts: a pooled analysis of five randomized clinical trials”. Circula-tion. vol. 106. 2002. pp. 3063-7.
10. McDaniel MC, Eshtehardi P, Sawaya FJ, Douglas JS Jr, Samady H. Contemporary clinical applications of coronary intravascular ultra-sound. JACC Cardiovasc Interv. 2011;4(11):1155-1167.
11. Predescu L.M et al. The value of invasive hemodynamic and imagistic evaluation to define the right PCI strategy. Romanian Journal of Car-diology Vol. 29, No. 2, 2019
12. Kassimis G, Raina T, Kontogiannis N, et al. How Should We Treat Heavily Calcified Coronary Artery Disease in Contemporary Practice? From Atherectomy to Intravascular Lithotripsy. Cardiovasc Re-vasc Med. 2019;20(12):1172-1183.
13. Predescu L.M, Zarma L, Bucsa A, Deleanu D, Ginghina C, et all Current treatment of left main coronary artery disease. Cor et Vasa 58 (2016) e328-e339.
14. Mohsen, Mohammed Khalil et al. “Stent fracture: how frequently is it recognized?.” Heart views : the official journal of the Gulf Heart Association vol. 14,2 (2013): 72-81;
15. Schömig A, Dibra A, Windecker S, Mehilli J, Suárez de Lezo J, Kaiser C, et al. A meta-analysis of 16 randomized trials of sirolimus-eluting stents versus paclitaxel-eluting stents in patients with coronary ar-tery disease. J Am Coll Cardiol. 2007;50:1373–80;
16. Chakravarty T, White AJ, Buch M, Naik H, Doctor N, Schapira J, et al. Meta-analysis of incidence, clinical characteristics and implications of stent fracture. Am J Cardiol. 2010;106:1075–80;
17. Doi H, Maehara A, Mintz GS, Tsujita K, Kubo T, Castellanos C, et al. Classification and Potential Mechanisms of Intravascular Ultrasound Patterns of Stent Fracture. Am J Cardiol. 2009;103:818–23.
 This work is licensed under a
This work is licensed under a