Andreea Varga1,2, Ioan Tilea1,2, Pietro Palermo3
1 George Emil Palade” University of Medicine, Pharmacy, Science and Technology, Targu Mures, Romania.
2 Department of Cardiology, IInd Clinic of Internal Medicine, Emergency Clinical County Hospital, Targu Mures, Romania
3 Centro Cardiologico Monzino, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS), Milan, Italy
Abstract: In patients presenting cardiovascular, pulmonary, muscular and metabolic diseases, exercise testing continues to be a predictive and diagnosis source of information. Diagnosis and assessment of coronary artery disease (known or suspected) encompass clinical and risk factors evaluation and well-defined non-invasive and invasive tests. Cardiopulmonary exercise test (CPET) is an evidence-based evaluation technique in Europe, USA and other countries. It consists in applying, directly supervised by a physician, a standardized incremental physiological stress (using cycle ergometer or treadmill) until the patient reaches the anaerobic threshold. CPET can assess the presence of myocardial ischemia, by three parameters: oxygen pulse, maximum oxygen uptake curve and elevation and relationship between maximum oxygen uptake variation and load variation. Furthermore, the CPET is useful in assessing the prognosis of cardiac patients, in choosing the most effective treatment, and in evaluating the response to treatment post-acute coronary syndrome, post-primary percutaneous coronary intervention or assessment of unexplained dyspnoea or myocardial ischemia in heart failure patients. CPET has a higher sensitivity, specifi city, positive predictive value and negative predictive value as compared to standard ECG stress testing in patients with suspected myocardial ischemia.
In this paper a brief review of current indications, analysis of standardized nine panel plot interpretation of CPET parameters related to myocardial ischemia are depicted.
Keywords: cardiopulmonary exercise test, indications, oxygen consumption, anaerobic threshold, myocardial ischemia.
List of abbreviations used in text
AT Anaerobic threshold
CAD Coronary artery disease
CPET (or CPX) Cardiopulmonary exercise test
CTEPH Chronic thromboembolic pulmonary hypertension
CV Cardiovascular
FEV1 Forced expiratory volume measured during the fi rst second of the forced breath
HF Heart failure
HR Heart rate
HR/VO2 Heart rate – Oxygen uptake relationship
IC Inspiratory capacity
MVV= FEV1x40 Maximal voluntary ventilation: volume of air expired in a specified period during repetitive maximal effort
PAH Pulmonary arterial hypertension
Peak VO2 Oxygen utilisation (mLO2 x kg-1x min-1)
PETCO2 End-tidal partial pressure of CO2 (mmHg)
PETO2 End-tidal partial pressures of O2 (mmHg)
PVOD Pulmonary Veno-Occlusive Disease
PTP Pretest probability
RC Respiratory compensation
RER Respiratory exchange ratio (respiratory quotient), RER=VCO2/VO2
SPTD Standard conditions: temperature 0 degrees Celsius, pressure 760 mmHg and dry
VC Vital capacity
VCO2 Carbon dioxide production/min corrected for STPD conditions
VD/VT Dead Space to Tidal Volume ratio
VE Ventilation
VE/VCO2 Minute ventilation/carbon dioxide production
VO2 Oxygen consumption/min corrected for STPD conditions
VO2/HR Oxygen pulse
VO2max Maximal O2 uptake
VT Tidal volume (mLO2 x kg-1 x min-1)
WR Work rate
∆VO2/∆WR Oxygen delivery (uptake)
INTRODUCTION
In patients presenting cardiovascular (CV), pulmonary or association of both diseases, exercise testing conti-nues to be a predictive and diagnosis source of infor-mation. Diagnosis and assessment of coronary artery disease (known or suspected) encompass clinical and risk factors evaluation and well-defined non-invasive and invasive tests.
Currently, non-invasive tests commonly used to diagnose the presence of coronary artery disease (CAD) can vary in sensitivity (45-97%) and specifi city (61-95%), accordingly to different authors1.
Recommendations for using non-invasive tools need to take into account the pre-test probability (PTP).1 In patients with stable angina symptoms and PTP at 15% to 65% intervals, an exercise ECG test could be used as an initial evaluation test. Electrocardiogram exer-cise testing has inherent limits described elsewhere, wide variability in reported sensitivity (45-50%) and specifi city (85-90%), respectively2,3. Cardiopulmonary exercise testing (abbreviated CPET or CPX) is a non-invasive diagnostic technique used to evaluate the physiologic response of the cardi-ovascular, pulmonary and muscle-energetic systems to an increase in physical stress, especially in cardiovas-cular and pulmonary disease suspected or diagnosed patients4,5. It is a distinct exercise testing that provides accurate and objective quantification of cardiorespira-tory fitness (peak VO2)6,7.
CPETs are used, and should be used more and more, with cardiac patients. Indeed, the CPET evalu-ates the functional capacity and helps to identify the cardiac, pulmonary or peripheral origin of a possible reduced functional capacity.
Furthermore, the CPET is useful in assessing the prognosis of cardiac patients, in choosing the most effective treatment, and in evaluating the response to treatment. The term “cardiac disease” comprises se-veral disorders that show typical and different pattern of gas kinetic during exercise. For this reason, it is important to know the specific typical behaviour of each cardiac disease before analysing the tests.
Using treadmill or electromagnetically braked cycle ergometer (preferred in most European countries), the progressive incremental workload exercise test (ramp) is frequently used for patients clinical evaluati-on; it is the most appropriate to obtain data about car-diac and pulmonary function, the functional capacity, the prognosis, and the response to therapy.
In the last two decades, applications of CPET are increasing, being used in diagnostic, appraising thera-peutic regimens and prognostic recommendations in heart failure (HF) patients, valvular diseases, hyper-trophic cardiomyopathy, pulmonary interstitial and vascular diseases. The use of CPET affords combined data illustrating exercise physiology in addition to non-invasive measurements (especially echocardiographic) of cardiac anatomy and function8. Table 1 outline the current indications of CPET.
The combined analysis of several parameters, which are obtained by incremental workload protocols, provides the most accurate information.
Data provided by the CPET relevant to gas exchan-ge measurements (ventilation versus CO2 output VE/VCO2 peak, analysis of O2 pulse and ∆VO2/∆WR slo-pe) can lead to identifying exercise-induced myocar-dial ischaemia, especially when conduction of rhythm disturbances are present at rest13-16.
In a recently published study, Belardinelli et al. pre-sent that in patients with chest pain, CPET indicates a better diagnostic and predictive accuracy than stan-dard exercise test in detection or rule-out obstructive CAD17.
Current guidelines for the management of stable CAD do not include CPET in the diagnostic algorithm of myocardial ischemia1.
A supervised CPET is recommended to prescribe an adapted physical activity and cardiac rehabilitation programme post-acute coronary syndrome, post-pri-mary percutaneous coronary intervention or assess-ment of unexplained dyspnoea or myocardial ischemia in HF patients18-20.
Nevertheless, in patients with diabetes mellitus use-fulness of CPET complementary to standard exerci-se testing needs further evidence, in order to assess functional capacity and exercise-induced ischemia, es-pecially when resting electrocardiogram is normal and interpretable8. Presence of anginal symptoms secondary to abnormalities of coronary artery tract where the presence of ischemia (ST-segment changes) can be assessed using CPET.
CARDIOPULMONARY EXERCISE NINE-PANEL PLOT
Using CPET to evaluate the gas exchange between the body cells and atmosphere through the interconnected cardiovascular and respiratory systems, can delineate the pathophysiology of performance restraint21. The nine-panel graphical array describes the cardiovascu-lar, ventilatory, ventilation-perfusion matching and metabolic responses to exercise. Starting from 1997, specifi c recommendations of graphical CPET panels, panels display and axes, were published. In 2017, Du-mitrescu and Rosenkranz presented an update on the graphical data display for clinical CPET22.
Panel 1 – VO2 and VCO2 gas kinetics
Panel 1 is the first panel to be observed in the iden-tification of possible exercise limitation. The VO2 and VCO2 gas kinetics analysis could enlighten a possible cardiovascular or respiratory limitation. In healthy subjects performing a 10 minutes CPET, there is a linear increment of workload to O2 consumption (VO2). Continuously to workout, VO2 increases at a standard rate of 10ml/min/W.22 Within the panel, the parameters and their position, slope, and linearity cor-respondence should be analysed: VO2, VCO2 versus time and work rate.
Panel 2 – Heart rate and oxygen pulse
The oxygen pulse (VO2/HR) defines the ratio of oxygen consumption to heart rate, inference of the VO2 normalized for the heart rate (HR). The oxygen pulse is a defined cardiac performance index, parti-cularly of the systolic output. In healthy subjects, the oxygen pulse increases as the workload increases and it reaches a plateau associated with a linear increase in HR. This pattern is a proper demonstration of a well-adjusted and adequate rise of the systolic output, correspondent to the HR and therefore, the cardi-ac output in response to exercise. A limitation of the systolic output is manifest in anaemia, hypoxemia or in the presence of muscle impairment.
Panel 3 – Respiratory equivalent. VO2 and
VCO2 relationship (V-slope). The first method of anaerobic threshold (AT) estimation
Heart rate and VCO2 calculated over VO2 in panel 3, graphically presents the heart frequency tendency end the relation of VO2 and VCO2. Plotting the VO2 and the VCO2 with axes using the same scale, allows cal-culating the anaerobic threshold (AT). Up to the AT, the VCO2 rises linearly to VO2. Subsequently to the AT, the VCO2 increases more rapidly and the slope of the curve depends on the degree of lactic acidosis buffering. The point of inflexion or the change of slope identifies the AT. The AT is reached anticipative in patients with altered cardiac function or muscle de-conditioning.
Panel 4 – Ventilatory equivalent for O2 and CO2 (VE/VO2 and VE/VCO2). The second method of AT estimation
This panel graphically presents the tendency of ven-tilatory equivalents, the ratio of VE to VO2 and VE to VCO2 versus time and work rate. The analysis of ventilatory equivalents provides information to the cli-nician on the presence of dead space/current volume ratio (VD/VT) alterations, concomitant to the venti-lation/perfusion ratio (V/Q) alterations. Furthermore, analysing the ventilatory equivalents, the AT point can be identified. VE/VO2 slope outline an initial reduc-tion followed by parallel direction to the work axis up to the AT. Therefore, it starts to increase related to ventilation increase, as a consequence of the CO2 necessary to the lactic acidosis tamponade. During the isocapnic tamponade period, on the other hand, the VE/VCO2 slope remains constant up to the respirato-ry compensation (RC) point. Beyond the point of RC also VE/VCO2 increases, and this is due to the parti-cular acidosis stimulus no longer compensated on the ventilatory drive.
Panel 5 – Ventilation
This panel presents the course of ventilation (L/min) during an increasing workload; an increase in venti-lation (change of slope of the curve) at the moment of workload above the anaerobic threshold (AT), is evident. The increase in the ventilatory drive is secon-dary to the increase in CO2, as a result of the lactic acidosis buffering and the increase in cellular metabo-lism. A second and further increase in ventilation is noticeable, which determines a second change in the curve slope, and a point of respiratory compensation is defi ned. At this moment the buffer systems are no longer able to compensate the pH, so that acidosis ap-pears, being a strong stimulation for hyperventilation. The phase of isocapnic tamponade defi nes the start phase of the anaerobic threshold up to the point of respiratory compensation. The next phase at the res-piratory compensation point is named uncompensated acidosis or respiratory compensation phase.
Panel 6 – Ventilatory efficacy
The VE/VCO2 report provides indications on the organism’s efficiency in eliminating the CO2 produced during the metabolic processes. The slope of the cur-ve defines the entity of ventilation increase, necessary to eliminate a given carbon dioxide production. A high slope value is associated with inefficient ventilation. There is no linearity of VE and VCO2 above the res-piratory compensation point. VE/VCO2 relationship is validated as a powerful prognostic parameter for cardiac and pulmonary disease22. The y-axis intercept derived from the chart is related to the dead space ventilation23.
Panel 7 – End-tidal partial pressures of O2 (PETO2, mmHg) and end-tidal partial pressure of CO2 (PETCO2, mmHg). The third method of AT estimation
The possible presence of ventilation/perfusion mis-match by decreased PETCO2 value is recognized in the ventilation/perfusion mismatch, and in case of hyper-ventilation. A concomitant increase of respiratory exchange ratio (panel 8) confi rms and identify a situa-tion of hyperventilation.
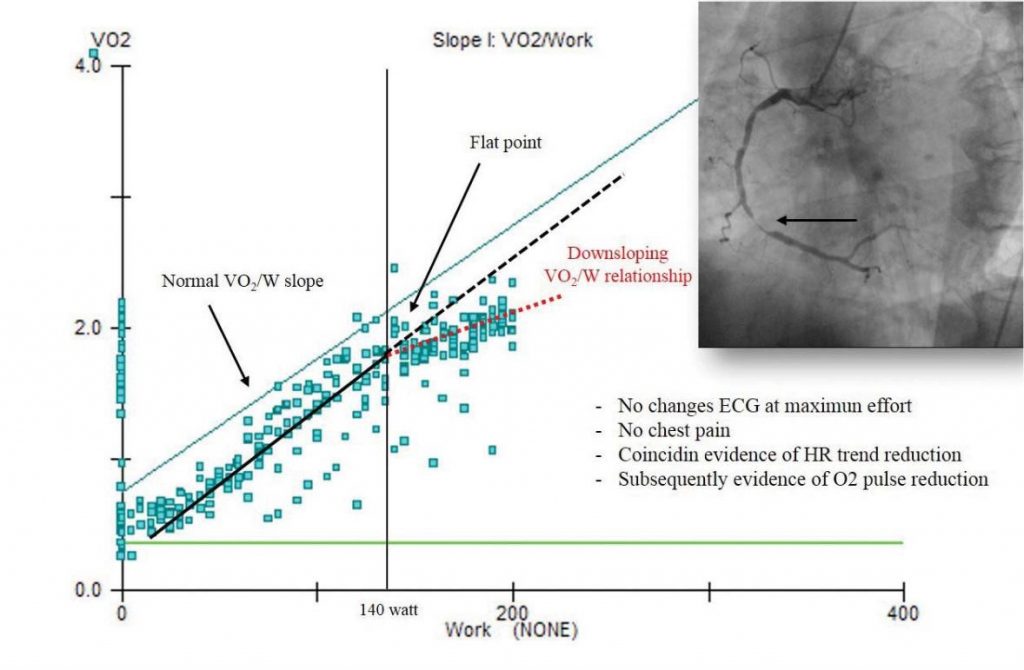
Figure 1. Presents CV, ventilatory, ventilation-perfusion matching and metabolic responses to exercise, displayed in standard nine-panel plot in a 51-year old athlete.
Panel 8 – Respiratory exchange ratio (respiratory quotient) (RER, R); RER=VCO2/ VO2
The metabolic pathway of chemical energy producti-on involves the use of oxygen for energy substrates oxidation (mainly carbohydrates and lipids) and CO2 produced by oxidative reactions. In the ATP produc-tion when fatty acids are used,23 O2 molecules are needed to oxidize a fatty acid molecule and 16 mo-lecules of CO2 are produced (RER=16/23=0.7) The carbohydrates oxidation involves the utilization of six O2 molecules and six molecules of CO2 are produced (RER=6/6=1). During CPET on the y-axis, RER begins at 0.8 and the rise above 1.0 validates a maximal exer-cise test21,24.
Panel 9 – Tidal volume, ventilation and respiratory frequency
The behaviour of ventilation is represented by the re-lation of tidal volume (VT) and ventilation (VE). The patient’s vital capacity (VC) and inspiratory capacity (IC) is pictured in a parallel line to the x-axis, and the maximum voluntary ventilation (MVV=FEV1x40) on a parallel line to the y-axis. The origin isogram deter-mines the ventilatory frequency. Breathing reserve is computed as MVV-VE using panel nine parameters24.
Figure 1. The 9 panel plot data display and inter-pretation. Cardiopulmonary exercise test of a 51-year old athlete. Ramp test with an increment of 20 W/Min. Nine-panel plot display of the results, with 10-second averaging. Panels marked in red (1,2,3): „Circulatory axis”, indicating that these panels show parameters important for the evaluation of circulato-ry function and circulatory reserve. Panels marked in green (1,5,9): „Ventilatory axis”, indicating that these panels show parameters important for the evaluation of ventilatory function and ventilatory reserve. Panels marked in blue (1,4,6,7): „Ventilatory efficiency axis”, indicating that these panels show parameters impor-tant for the evaluation of ventilatory efficiency and the relationship of ventilation to perfusion during exercise. Panels marked in grey (8): „General metabolic changes” (RER).
(Courtesy of Dr. med Daniel Dumitrescu, Klinik für Allgemeine und Interventionelle Kardiologie/Herz-und Diabeteszentrum Nordrhein-Westfalen, Germany)
FUNCTIONAL CAPACITY LIMITATION
Different cardiac, ventilatory, pulmonary or pe-ripheric vessel abnormalities can induce exercise li-mitations. CPET focuses on a number of questions related to physiology and pathophysiology workout, though identification of the unique cause of limitation is complex.
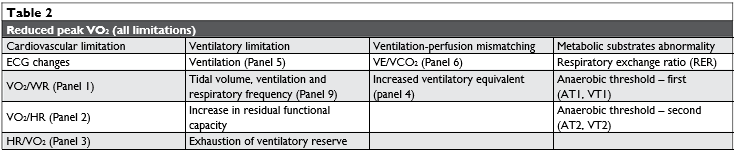
By equating the CPET derived parameters, panel analyse, it becomes feasible to identify disorders un-derlying abnormalities24,25 (Table 2).
CPET TO ASSESS MYOCARDIAL ISCHEMIA
Systematize clinical exercise test interpretation re-lays on key CPET variables for diagnostic and prognos-tic utility in patients with ischemic heart disease. The use of CPET to detected cardiovascular dysfunction induced by mild ischemia was not conclusive in previ-ous studies; still, changes in specific parameters were identified as a response at incremental workload in extended myocardial ischemia.26 Several case reports and research papers revealed the same exercise-indu-ced left ventricular dysfunction detected by CPET, in both microvascular and macrovascular coronary ar-tery disease (CAD)27,28. In particular cases, if the ECG is not accurate for myocardial ischemia (i.e. intraven-tricular blocks, pacemakers) gas exchange analysis can provide valuable information on O2 consumption and O2 supply imbalance in mitochondria17.
Concomitant use of complementary functional methods with current cardiac imaging studies de-monstrated a blunt value in the diagnosis of myocar-dial dysfunction, secondary to non-obstructive coro-nary artery disease in symptomatic patients. CPET can detect cardiac dysfunction using specific sets of valu-es identifying the abnormalities in stroke volume and heart rate in this defined subset of CAD29.
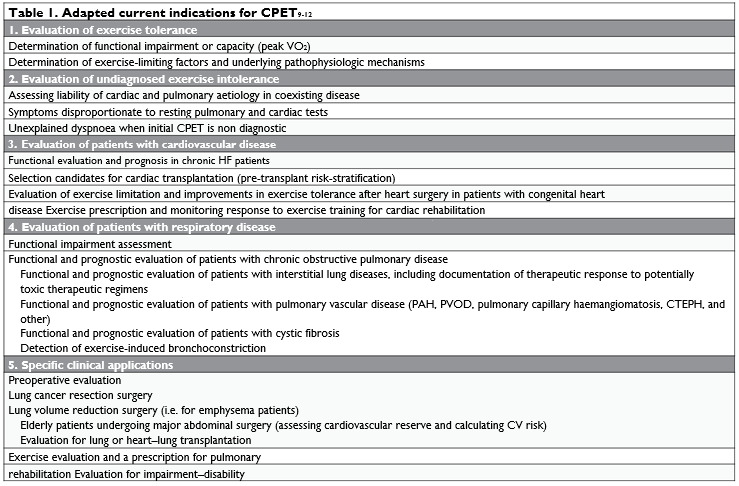
CPET determinants to detect myocardial ischemia: O2 pulse, VO2 slope and VO2/Watts.
Maximal O2 uptake (VO2max) – Panel 1
In different population, age, sex, fitness exercise level, weight, height, ethnicity and genetic variability influen-ce the average values of VO2max. Depending on these factors, different equations for predicting the standard values of VO2max have been determined, although the equation proposed by Wasserman and Whipp is the most used24. In performing exercise test, VO2 raises at a rate of 10mL/min/W. Maximal oxygen uptake (VO2max) represents the highest O2 consumption possible to at-tained during an incremental exercise test as proven by the fall of VO2 to raise linearly to increasing work rate.
Peak VO2 – Panel 1
Peak oxygen uptake (peak VO2) represents the highest O2 consumption achieved during presumed maximal effort in an incremental exercise; it can equal the VO2max.
As a marker of cardiovascular limitation, in a Finnish population, Khan et al. concluded that maximal O2 uptake (peak VO2) reduction is directly associated with non-fatal myocardial infarction and heart failure events30. Preserving a high peak VO2 was demonstra-ted as a protective prognosis value31.
Work rate – VO2 to watt, (VO2/WR) – Panel 1
Abnormal pattern, reduction or non-linear relation-ship of work rate in a standardized CPET represents the first signs of exercise limitation, and indices of left ventricular restraint in case of myocardial ischemia. The VO2/Work rate presents the coupling of external to cellular respiration, graphically represented by the ability of O2 consumption to track the workout incre-ase. The ∆VO2/∆W ratio follows a standard course along with the low workload, with a straight decline consecutive to myocardial ischemia inducing myocar-dial dyskinesis at the AT point. The VCO2 progress increases abruptly as the VO2 continues to decline24. There is also a documented correlation of the ECG changes at the moment or subsequently to ∆VO2/∆W decrease.16 The study of Popovic and collab. validate the ∆VO2/∆W recumbent cycle-ergometer CPET as an evident additional value in the prognosis of CAD32.
Oxygen pulse (VO2/HR), reduce response – Panel 2
Oxygen pulse (oxygen consumption and heart rate, VO2/HR, mL/heartbeat) and stroke volume (mL/heart-beat, blood volume ejected to the aorta during the systole) are both parameters indicator of left ventricle performance during workload. Decline or fl attening of the VO2/HR curve plotted over workload time indica-te the oxygen pulse and stroke volume reduction, as a subtle indicator of myocardial ischemia induced by left ventricular dysfunction26,33. Chaudry et al. tested the new mathematical model DHR-WR slope, expressing the changes in HR slope in late exercise plotted to the workload, demonstrating an increased sensitivity and negligible specificity reduction of the undertreated atherosclerosis diagnosis34.
Heart rate – Oxygen uptake (HR/VO2) relationship – Panel 3 (V-slope in AT determination)
Progressively increase and concomitant with altering of the linearity of HR to VO2, will be observed in the panel, in myocardial ischemia pattern22.
CONCLUSION
CPET demonstrated the role in CAD assessment. Particular CPET parameters and panel analyse, extend possibilities of myocardial ischemia detection outside the limits of using more expensive, harmful and inva-sive traditional investigating procedures. In patients with suspected myocardial ischemia, CPET has a hi-gher sensitivity, specificity, positive predictive value and negative predictive value as compared to standard ECG stress testing.
Conflict of interest: none declared.
Acknowledgements: This paper is partially suppor-ted by the University of Medicine, Pharmacy, Science and Technology of Târgu Mureş Research Grant num-ber 615/8/17.01.2019.
All authors contributed equally to this paper and sho-uld be viewed as first authors.
References
1. Task Force Members, Montalescot G, Sechtem U, Achenbach S, An-dreotti F, Arden C, Budaj A, Bugiardini R, Crea F, Cuisset T, Di Ma-rio C, Ferreira JR, Gersh BJ, Gitt AK, Hulot JS, Marx N, Opie LH, Pfisterer M, Prescott E, Ruschitzka F, Sabaté M, Senior R, Taggart DP, van der Wall EE, Vrints CJ; ESC Committee for Practice Guide-lines, Zamorano JL, Achenbach S, Baumgartner H, Bax JJ, Bueno H, Dean V, Deaton C, Erol C, Fagard R, Ferrari R, Hasdai D, Hoes AW, Kirchhof P, Knuuti J, Kolh P, Lancellotti P, Linhart A, Nihoyan-nopoulos P, Piepoli MF, Ponikowski P, Sirnes PA, Tamargo JL, Ten-dera M, Torbicki A, Wijns W, Windecker S; Document Reviewers, Knuuti J, Valgimigli M, Bueno H, Claeys MJ, Donner-Banzhoff N, Erol C, Frank H, Funck-Brentano C, Gaemperli O, Gonzalez-Juanatey JR, Hamilos M, Hasdai D, Husted S, James SK, Kervinen K, Kolh P, Kris-tensen SD, Lancellotti P, Maggioni AP, Piepoli MF, Pries AR, Romeo F, Rydén L, Simoons ML, Sirnes PA, Steg PG, Timmis A, Wijns W, Windecker S, Yildirir A, Zamorano JL. 2013 ESC guidelines on the management of stable coronary artery disease. The Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013;34(38):2949-3003.
2. Froelicher VF, Lehmann KG, Thomas R, Goldman S, Morrison D, Edson R, Lavori P, Myers J, Dennis C, Shabetai R, Do D, Froning J. The electrocardiographic exercise test in a population with reduced workup bias: diagnostic performance, computerized interpretation, and multivariable prediction. Veterans Affairs Cooperative Study in Health Services #016 (QUEXTA) Study Group. Quantitative Exer-cise Testing and Angiography. Ann Intern Med. 1998;128:965-74.
3. Morise AP, Diamond GA. Comparison of the sensitivity and specific-ity of exercise electrocardiography in biased and unbiased popula-tions of men and women. Am Heart J. 1995;130:741-47.
4. Gibbons RJ, Balady GJ, Beasley JW, Bricker JT, Duvernoy WF, Froeli-cher VF, Mark DB, Marwick TH, McCallister BD, Thompson PD Jr, Winters WL, Yanowitz FG, Ritchie JL, Gibbons RJ, Cheitlin MD, Ea-gle KA, Gardner TJ, Garson A Jr, Lewis RP, O’Rourke RA, Ryan TJ. ACC/AHA Guidelines for Exercise Testing. A report of the Ameri-can College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on Exercise Testing). J Am Coll Cardiol. 1997;30:260-311.
5. Ross A. Exercise testing. In ACSM’S Guidelines for Exercise Testing and Prescription. Pescatello LS, Arena R, Riebe D, Thompson PD. 9th edn. 2014.Wolters Kluwer/Lippincott Williams & Wilkins, Phila-delphia, USA.
6. Guazzi M, Adams V, Conraads V, Halle M, Mezzani A, Vanhees L, Arena R, Fletcher GF, Forman DE, Kitzman DW, Lavie CJ, Myers J; European Association for Cardiovascular Prevention & Rehabili-tation; American Heart Association. EACPR/AHA Scientific State-ment. Clinical recommendations for cardiopulmonary exercise testing data assessment in specific patient populations. Circulation 2012;126(18):2261-74.
7. Guazzi M, Arena R, Halle M, Piepoli MF, Myers J, Lavie CJ. 2016 focused update: clinical recommendations for cardiopulmonary ex-ercise testing data assessment in specifi c patient populations. Eur Heart J. 2018;39(14):1144-61.
8. Guazzi M, Bandera F, Ozemek C, Systrom D, Arena R. Cardio-pulmonary Exercise Testing: What Is its Value? J Am Coll Cardiol. 2017;70(13):1618-36.
9. American Thoracic Society; American College of Chest Physicians. ATS/ACCP Statement on cardiopulmonary exercise testing. Am J Respir Crit Care Med. 2003;167(2):211-77.
10. Albouaini K, Egred M, Alahmar A, Wright DJ. Cardiopulmonary ex-ercise testing and its application. Postgrad Med J. 2007;83(985):675-82.
11. ERS Task Force, Palange P, Ward SA, Carlsen KH, Casaburi R, Galla-gher CG, Gosselink R, O’Donnell DE, Puente-Maestu L, Schols AM, Singh S, Whipp BJ. Recommendations on the use of exercise testing in clinical practice. Eur Respir J. 2007;29(1):185-209.
12. Leuchte HH, Ten Freyhaus H, Gall H, Halank M, Hoeper MM, Kaem-merer H, Kähler C, Riemekasten G, Ulrich S, Schwaiblmair M, Ewert R. Risk stratification strategy and assessment of disease progression in patients with pulmonary arterial hypertension: Updated Recom-mendations from the Cologne Consensus Conference 2018. Int J Cardiol. 2018;272S:20-9.
13. Popovic D, Guazzi M, Jakovljevic DG, Lasica R, Banovic M, Osto-jic M, Arena R. Quantifi cation of coronary artery disease using dif-ferent modalities of cardiopulmonary exercise testing. Int J Cardiol. 2019;285:11-3.
14. Mazaheri R, Shakerian F, Vasheghani-Farahani A, Halabchi F, Mirsha-hi M, Mansournia MA. The usefulness of cardiopulmonary exercise testing in assessment of patients with suspected coronary artery dis-ease. Postgrad Med J. 2016;92(1088):328-32.
15. De Lorenzo A, da Silva CL, Souza FCC, Serra S, Marino P, Sl Lima R. Clinical, scintigraphic, and angiographic predictors of oxygen pulse abnormality in patients undergoing cardiopulmonary exercise test-ing. Clin Cardiol. 2017;40(10):914-8.
16. Belardinelli R, Lacalaprice F, Carle F, Minnucci A, Cianci G, Perna G, D’Eusanio G. Exercise-induced myocardial ischaemia detected by cardiopulmonary exercise testing. Eur Heart J. 2003;24(14):1304-13.
17. Bardinelli R, Lacalaprice F, Tiano L, Muçai A, Perna GP. Cardiopul-monary exercise testing is more accurate than ECG-stress testing in diagnosing myocardial ischemia in subjects with chest pain. Int J Car-diol. 2014;174(2):337-42.
18. Pavy B, Iliou MC, Vergès-Patois B, Brion R, Monpère C, Carré F, Aeberhard P, Argouach C, Borgne A, Consoli S, Corone S, Fisch-bach M, Fourcade L, Lecerf JM, Mounier-Vehier C, Paillard F, Pierre B, Swynghedauw B, Theodose Y, Thomas D, Claudot F, Cohen-Sol-al A, Douard H, Marcadet D; Exercise, Rehabilitation Sport Group (GERS); French Society of Cardiology. French Society of Cardiology guidelines for cardiac rehabilitation in adults. Arch Cardiovasc Dis. 2012;105:309-28.
19. Piepoli MF, Corrà U, Benzer W, Bjarnason-Wehrens B, Dendale P, Gaita D, McGee H, Mendes M, Niebauer J, Zwisler AD, Schmid JP. Secondary prevention through cardiac rehabilitation: from knowl-edge to implementation. A position paper from the Cardiac Re-habilitation Section of the European Association of Cardiovascu-lar Prevention and Rehabilitation. Eur J Cardiovasc Prev Rehabil. 2010;17(1):1-17.
20. Marcadet DM, Pavy B, Bosser G, Claudot F, Corone S, Douard H, Iliou MC, Vergès-Patois B, Amedro P, Le Tourneau T, Cueff C, Ave-dian T, Solal AC, Carré F. French Society of Cardiology guidelines on exercise tests (part 2): indications for exercise tests in cardiac diseases. Arch Cardiovasc Dis. 2019;112(1):56-66.
21. Guazzi M, Arena R, Halle M, Piepoli MF, Myers J, Carl J. Lavie CJ. 2016 focused update: clinical recommendations for cardiopulmonary exercise testing data assessment in specific patient populations. Eur Heart J. 2018;39(14):1144-61.
22. Dumitrescu D, Rosenkranz S. Graphical data display for clinical car-diopulmonary exercise testing. Ann Am Thorac Soc. 2017;14(1):S12-S21.
23. Gargiulo P, Apostolo A, Perrone-Filardi P, Sciomer S, Palange P, Agostoni P. A non-invasive estimate of dead space ventilation from exercise measurements. PLoS One 2014;9(1):e87395.
24. Wasserman K, Hansen J, Sue D, Stringer W, Sietsema KE, Sun XG, Whipp VJ. Principles of Exercise Testing and Interpretation, 5th edn. 2012. Lippincott Williams & Wilkins, Philadelphia, USA.
25. Pinkstaff S, Peberdy MA, Fabiato A, Finucane S, Arena R. The clini-cal utility of cardiopulmonary exercise testing in suspected or con-firmed myocardial ischemia. Am J Lifestyle Med. 2010;4:327-348.
26. Munhoz EC, Hollanda R, Vargas JP, Silveira CW, Lemos AL, Hol-landa RM, Ribeiro JP. Flattening of oxygen pulse during exercise may detect extensive myocardial ischemia. Med Sci Sports Exerc. 2007;39(8):1221-6.
27. Chaudhry S, Arena R, Wasserman K, Hansen JE, Lewis GD, Myers J, Belardinelli R, Brian LaBudde B, Menasco N, Boden WE. The utility of cardiopulmonary exercise testing in the assessment of suspected microvascular ischemia. Int J Cardiol. 2011;148:e7-e9
28. Chaudhry S, Arena R, Wasserman K, Hansen JE, Lewis GD, My-ers J, Chronos N, Boden WE. Exercise-induced myocardial isch-emia detected by cardiopulmonary exercise testing. Am J Cardiol. 2009;103(5):615-9.
29. Chaudhry S, Arena R, Bhatt DL, Verma S, Kumar N. A practical clinical approach to utilize cardiopulmonary exercise testing in the evaluation and management of coronary artery disease: a primer for cardiologists. Curr Opin Cardiol. 2018;33(2):168-77.
30. Khan H, Jaffar N, Rauramaa R, Kurl S, Savonen K, Laukkanen J. Car-diorespiratory fitness and nonfatal cardiovascular events: A popula-tion-based follow-up study. Am Heart J. 2017;184:55-61.
31. Al-Mallah MH, Sakr S, Al-Qunaibet A. Cardiorespiratory fi tness and cardiovascular disease prevention: An update. Curr Atheroscler Rep. 2018;20(1):1.
32. Popovic D, Guazzi M, Jakovljevic GD, Lasica R, Banovic M, Osto-jic M, Arena R. Quantification of coronary artery disease using dif-ferent modalities of cardiopulmonary exercise testing. Int J Cardiol. 2019;285:11-3.
33. Balady GJ, Arena R, Sietsema K, Myers J, Coke L, Fletcher GF, For-man D, Franklin B, Guazzi M, Gulati M, Keteyian SJ, Lavie CJ, Macko R, Mancini D, Milani RV; American Heart Association Exercise, Car-diac Rehabilitation, and Prevention Committee of the Council on Clinical Cardiology; Council on Epidemiology and Prevention; Coun-cil on Peripheral Vascular Disease; Interdisciplinary Council on Qual-ity of Care and Outcomes Research. Clinician’s Guide to cardiopul-monary exercise testing in adults: a scientific statement from the American Heart Association. Circulation 2010;122(2):191-225.
34. Chaudhry S, Kumar N, Behbahani H, Bagai A, Singh BK, Menasco N, Lewis GD, Sperling L, Myers J. Abnormal heart-rate response dur-ing cardiopulmonary exercise testing identifies cardiac dysfunction in symptomatic patients with non-obstructive coronary artery disease. Int J Cardiol. 2017;228:114-21.
 This work is licensed under a
This work is licensed under a