Download PDF
https://doi.org/10.47803/rjc.2020.30.4.553
Dan Musat1, Suneet Mittal1
1 Department of Cardiovascular Services, Snyder Center for Comprehensive Atrial Fibrillation, Valley Health System, Paramus, NJ
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia in the current era, affecting 43.6 million people worldwide in 2016 (about 0.5 % of global population)1; a more than 30% increase over the past 20 years2. Future predictions suggest an worldwide increase in AF burden of more than 60% by 2050.2 This imposes a tremendous burden on healthcare systems worldwide with an estimated annual cost up to $ 26 billion in USA3 and € 3.3 billion in Europe4. AF has a progressive natural history, from paroxysmal AF (episodes lasting less than 7 days and terminated either spontaneously or after interventions), to persistent AF (episodes lasting more than 7 days), and eventually to permanent AF if no efforts are made to restore and maintain sinus rhythm1. AF management strategies include either a rate or a rhythm control, depending on the symptoms and patient preference, using medical treatment such as AV node blocking agents or antiarrhythmic medication or a more invasive approach with surgical ablation, percutaneous catheter ablation or a hybrid approach. The efficacy of any intervention for AF is judged by the absence of any recurrence of AF lasting 30 seconds or longer, an endpoint chosen by EHRA/ACC/ AHA/HRS inter-societal convention1,5,6. This arbitrary, short duration endpoint has sparked many controversies7,8, however it has been the gauge by which any new techniques, tools and technologies have been evaluated throughout the years. Multiple randomized trials9-12, non-randomized series and meta-analyses have demonstrated that AF catheter ablation portends a superior success to anti-arrhythmic medication for maintaining of sinus rhythm and preventing AF recurrences13. The long term success of AF ablation is significantly improved with control of comorbidities, like diabetes, hypertension, hyperlipidemia, and lifestyle modifications including evaluation and treatment of sleep apnea, weight management (with a goal of ≥10% of weight loss and a BMI below 27 kg/m2), increase in fitness and alcohol use reduction or cessation, as well as smoking cessation1.
SURGICAL ATRIAL FIBRILLATION ABLATION
The race for AF treatment has started since early 1980’s with cardiac surgeons leading the way. Although the AF pathophysiology was not very well understood at that time, the goal was to slice the atria to decrease the area where AF wavelets could form reentrant circuits and to isolate the “hollow structures” in the atria (cava veins, pulmonary veins and left atrial appendage)14. Dr. James Cox was the pioneer in development of the technique for Cox-Maze procedure, which involves multiple cut and sew lesions of the left and right atrium. The procedure went through a series of iterations and evolved in widely used Cox-MAZE IV15,16 (Figure 1), using cut and sew lesions, along with radiofrequency and cryo lesions, to create electrical isolation of the pulmonary veins and posterior wall, as well as left atrial appendage excision. Long-term results have been reported over the years in multiple series of hundreds of patients17, one of the largest being a series by Khiabani et al. which reported outcomes of 853 patients undergoing Cox-Maze IV procedure, 60% having non-paroxysmal AF and about a quarter who failed a previous catheter ablation. They showed freedom from atrial tachyarrhythmias, off antiarrhythmic medication, of 84%, 71% and 61% at 1 year, 5 years and 10 years respectively16. Although the long-term results are very attractive, this invasive AF treatment option as standalone procedure is not appealing, so most of the times is performed as concomitant procedure at the time of open-heart surgery for other reasons. The associated risks are those of open heart surgeries with a perioperative mortality of 0.74% – 3.3% and overall complications rates ranging from 3.2 to 21% with a higher need for pacemaker therapy, ranging from 1.03% up to 12% in different series16-20. This approach in long term follow-up, retrospective analyses, have suggested an improved mortality and lower stroke rates17,18,21. Advances in the ablation tools and technologies led to development of minimally invasive surgical techniques for off pump epicardial or hybrid epicardial/endocardial ablation (Figure 1), however the results suggest a lower long term success rate 62-83%20,22, likely due to inferior lesion durability, and a higher complication rate, mostly conversion to sternotomy and bleeding complications20,22,23. Improvement of the convergent hybrid procedure technique led to a significant decline in complication, and with epicardial posterior wall isolation, endocardial PVI and more recently left atrial appendage exclusion, offer a more appealing mini-invasive approach mimicking the Cox-Maze IV22, especially in patients with persistent or longstanding persistent AF. Overall, surgical AF ablation has shown to have a higher efficacy than endocardial catheter AF ablation in long term maintenance of sinus rhythm22,24.
MINIMALLY INVASIVE ATRIAL FIBRILLATION CATHETER ABLATION
The era for less invasive ablation procedures started with introduction of catheter based ablation by Dr. Melvin Scheinman in 1982, born out as necessity for a treatment solution in patients who failed medical treatment and were not candidates for surgical approach. He initially used high-energy (300J – 500J) direct current shocks to ablate the fast AV nodal pathway for treatment of supraventricular tachycardia25,26 and to control irregular cardiac rhythm in patients with AF (Figure 2). This transitioned into unipolar radiofrequency ablation, introduced by SK Huang in 198727. The new technology provided the tools to attempt of reproducing the surgical ablation with a more appealing mini invasive approach. There were several series published that have used various linear ablation approaches in the left and/or right atrium, with success ranging from 33-79% in maintaining sinus rhythm at one year28-31. However, these approaches were plagued by long procedural and fluoroscopy duration and high complication rates up to 30 %31. The revolutionary discovery in 1997 by Dr. Michel Haissaguerre that the impulses responsible for starting AF originate in the pulmonary veins (Figure 2) has opened the current era for development of minimally invasive techniques and technologies for catheter based treatment of AF32,33. In the series of 45 patients, he reported that 94% of the ectopic foci starting AF were detected in the pulmonary veins (PV)32. Catheter ablation has become the method of choice for rhythm control strategy of symptomatic paroxysmal or persistent AF, currently recommended either as first choice or after failure of an antiarrhythmic medication1. During early experience, catheter manipulation and positioning guided only by fluoroscopy and contrast angiograms resulted in high fluoroscopy time and radiation exposure. In the late 1990’s the non-fluoroscopy tridimensional (3D) mapping systems started their development34. They are providing the possibility of reconstructing intracardiac and vascular anatomy, are capable of merging with pre-procedural cardiac imaging as CT or MRI, and most important they provide electro-anatomic activation and substrate maps with the ability to visualize the mapping and ablation catheters. Over the years, with technological advances, the 3D maps have become extremely accurate, currently being the standard of care for vast majority of ablations, with substantial decrease in fluoroscopy time and radiation exposure and an increase in success and safety of ablations35. In association with intracardiac echocardiogram, feedback about tissue contact provided by the newer ablation catheters, an increasing number of centers nowadays are performing non-fluoroscopic AF catheter ablations36-38. The initial experience with ablation of the induced pulmonary vein trigger foci inside the veins has encountered a significant occurrence in pulmonary vein stenosis ranging from 3-42%, with high AF recurrence rates32,39,40. Recognition of a high prevalence, in up to 69% of patients, of multiple arrhythmogenic veins led to an empiric all pulmonary vein isolation (PVI) approach40. This approach addresses the triggers for AF initiation, by disconnecting electrically the veins from the left atrium, as well as neuro-modulation factors by affecting the ganglionic plexi usually found at the antrum of pulmonary veins41,42. It has become the cornerstone of the AF ablation, the most popular approaches being segmental ostial ablation of the muscle sleeve connections and linear lesions around the antrum pulmonary veins (wide area circumferential ablation (WACA)), using either point-by-point radio-frequency applications or single-shot solutions1. Various types of energy have been considered over time for PVI, currently widely used being radiofrequency and cryothermal energies, with newer ones as pulse field electroporation and ultra-low cryothermal energy being in the early stages of development.
RADIOFREQUENCY ABLATION
Radiofrequency (RF) has been the first and it still remains the preferred type of energy used for AF ablation. RF energy is delivered in a unipolar fashion using alternative current, at a frequency of about 550 kHz, between the tip of the ablation catheter inside the heart and a dispersive grounding pad on the surface of the skin43. The lesion is created by thermal destruction of the tissue through resistive heating at the site of contact of the catheter to the tissue surface (up to 1 mm deep) and via passive conductive heating of the deeper tissue from the superficial layers43,44. In order to obtain an adequate lesion depth, due to low conductivity of the heat in the myocardial tissue, the RF application needs to be 20-60 sec long to reach maximal temperature inside the tissue and thermal equilibrium (constant heat flow from electrode to the tissue) to achieve the deepest lesion45. Operators have to be aware of “thermal latency” after RF delivery is stopped, as conductive heat continues to travel through the tissue and increase the temperature, thus possible affecting sensitive tissues (AV node area, es-ophageal tissue)45. A temperature above 50°C results in irreversible damage of the myocardial tissue due to coagulation and destruction of the cells and structural network43,46. Various factors affect the lesion size greater tissue contact and force of contact, larger catheter size, higher power, longer duration and higher electrode temperature lead to larger and deeper lesions43.
POINT-BY POINT RADIOFREQUENCY ABLATION
The first type of catheter used for AF ablation was non-irrigated and as data started to accumulate it was clear this was not an ideal tool, creating non-transmural lesions, associated with high recurrence rates of AF due to PV – left atrium (LA) reconnections47. In addition, high temperatures at the tissue contact (>100°C) would result in charring (deposits of denaturated plasma proteins) on the tip of the catheter leading to impedance rises, inefficient RF delivery and potential for thrombus formation and embolization43,46. These shortfalls of non-irrigated catheters led to development of the open irrigation catheters that currently are the norm for AF ablation. During RF delivery, the tip of the ablation catheter is constantly flushed via small ports with normal saline. This provides the ability to deliver higher power without increase in impedance or char formation, resulting in deeper, transmural lesions. However, the local tissue temperature feedback is lost, leading to possible com-plications due to excessive tissue heating and “steam pops” from boiling fluids inside the tissues45,46. Although the success of AF ablations improved47 with open irrigation catheters, long term results of AF ablations were far from perfect and it was recognized that the Achile’s heel of RF ablation remains the inability to provide efficient and durable lesions, with up to 70% of pulmonary vein reconnections1,48. Surrogate markers, as baseline impedance, impedance change, catheter position on reconstructed 3D maps proved to be inadequate49,50. Therefore, the efforts have focused to develop the tools for better evaluation of catheter stability and contact to the tissue during RF application, to assure transmural and contiguous lesions. The need to provide catheter-tissue contact feedback to the operator during RF application, led to development of contact force (CF) catheters. Two different contact force technologies have emerged. One platform uses a small spring that connects the ablation tip electrode to the catheter shaft, and translates the degree of this spring microdeflections into CF (Thermocool Smartouch CF, Biosense Webster Inc., Diamond Bar, California, USA). The other platform uses three optical fibers to convert the microdeformation of a deformable body inside the catheter tip into CF (TactiCath, Endosense/St Jude Medical, St Paul, Minnesota, USA). In TactiCath Contact Force Ablation Catheter Study (TOCCATA) Study and subsequent analyses49,51,52 it was demonstrated that a CF target of >10 g provides an adequate lesion, with 100% of AF recurrence when the RF lesions were applied with CF <10 g and 20% AF recurrence when lesions were performed with a CF ≥20 g51. There was a wide variability in CF between the operators, with higher success when higher CF was applied consistently51,52 and good stability of the catheter48,53. Similar findings were reported in SMART-AF trial50 using Thermocool Smartouch catheter with improved ablation success when the CF >18 g and when the operators maintained ≥80% of the time CF in the prespecified range, suggesting that besides the CF, stability plays an important role for durable lesion formation. Safety concerns were raised for both catheters when a CF >40 g, with increase in the incidence of tamponade50,51. Although the initial data was encouraging, with observational and some randomized studies suggesting a significant improvement in the success of AF ablations (71% vs 61%, especially for paroxysmal AF), decrease in the procedural, fluoroscopy and in RF times54-57, the initial enthusiasm vanished as more data accumulated. A systematic review and meta-analysis on almost 10,000 patients who were part of 26 observational and 9 randomized studies comparing CF ablation catheters with non-CF catheters showed that overall analysis of all the studies reproduced initial findings. However, when the analysis was restricted to the randomized studies only, the benefits of CF-guided ablation in terms of safety, efficacy, and procedural characteristics were no longer significant58. This was thought to be in part due to participation in the trials of experimented operators, already vexed with catheters without CF feedback, as well as less experimented operators with high variation in adequate CF maintenance during RF applications59. The race for better ablation tools with more effective and durable PV isolation has reached a new stage with the newest catheters and software capable of integrating multiple ablation variables to evaluate the efficacy of ablation and tissue characteristics during RF application. Ablation index (AI) represents a novel marker of ablation lesion quality developed and integrated into the automated lesion tagging software (Vi-siTag) in the CARTO 3 3D electroanatomic mapping system (Biosense Webster, Inc, Diamond Bar, CA) that incorporates power, CF, and time, in a proprietary weighted formula and was found to accurately estimate ablation lesion size60,61. An AI value of more than 480-540 at the anterior and roof regions and 370-380 at the posterior regions were predictive of freedom from acute and long term reconnections respectively60,62. Currently accepted targets are 550 for anterior and 400 for posterior regions60,61. A meta-analysis comparing AI guided procedures with non-AI guided approach, in 2306 patients enrolled in eleven studies63, showed a significant shorter procedural time (141.0 vs. 152.8 min, P = 0.01), shorter ablation time (21.8 vs. 32.0 min, P < 0.00001), higher first-pass isolation (93.4% vs. 62.9%, P < 0.001) and lower rates of acute PV reconnections (18.0% vs 35.0%; P = 0.006). In follow-up, atrial arrhythmia recurrence was also significantly lower in AI compared to non-AI catheter ablation (11.8% vs. 24.9%, P = 0.0003). Besides the lesion size and depth, for point-by-point ablation techniques, lesion contiguity has been recognized to play an important role for PV recon-nections. When an interlesion target was ≤6 mm, in the ‘CLOSE’-protocol study, the incidence of acute, adenosine proof isolation, was higher than conventional AI guided ablation (97% vs 82% p < 0.001)61. Freedom from atrial tachyarrhythmias at 12 months after single-procedure was 94% in ‘CLOSE’ vs. 80% in conventional group (P < 0.05). The vast majority of re-connections at repeat ablations were associated with either interlesion distance >6 mm and/or AI <400/550 (100% vs. 83%, P = 0.99 between ‘CLOSE’ and conventional groups respectively). Other innovations in lesion optimization were introduced with Boston Scientific IntellaNav MIFI™ Open-Irrigated catheter (Boston Scientific, Marlborough, MA, USA) which has in its tip 3 mini-electrodes capable of providing local bipolar electrograms and in conjunction with the 3D RHYTHMIA HDx™ (Boston Scientific, Marlborough, MA, USA) mapping system offers local and real time information about lesion formation. With ablation, mini-electrodes EGMs show a more substantial reduction in local amplitude compared to common bipoles. Mini-electrode guided ablation, to maximal EGM attenuation, results in 91% transmural lesions (similar to standard 60 seconds ablation) but with a significant reduction in ablation time (23.4 ± 7.8 seconds) and potentially avoiding extracardiac injuries64. This has improved further with the introduction of DirectSense™ (Boston Scientific, Marlborough, MA, USA) technology, capable of measuring the local tissue impedance with a superior identification of type of local tissue and a much better correlation of local impedance change with lesion formation when compared with transthoracic impedance65-67. The technology is based on a non-stimulatory alternating current (5.0 A at 14.5 kHz) delivered between the tip and the proximal ring to create a local potential field, with the mini-electrodes within the tip measuring potential field distortions caused by nearby cardiac structures or contact with high-resistivity myocardium65-67. In CHA-RISMA pilot trial65 a local impedance decrease during RF application of ≥15 Ω was strongly associated with an effective and durable lesion, irrespective of the baseline voltage of the site, with 91% atrial arrhythmia free at a mean follow-up of 404 ± 111 days. A local impedance decrease of more than 30 Ω has raised concerns for “steam-pops”. The ability for meaningful feedback about lesion formation using these new technologies has opened the appealing strategies of high power and short duration ablation, with shorter procedure and ablation times, better lesion formation and contiguity, with promising increased safety for extracardiac structures as well68-70. A new catheter, QDOT Micro catheter (Biosense Webster, Inc., Irvine, California) capable to deliver RF lesions with a 90W power showed promising results in the first in humans’ series of 52 patients with pa-roxysmal AF71. The high power RF was applied for 4 seconds per lesion, with 100% acute pulmonary vein isolation with study catheter and demonstrated good safety profile with only 2 complications (1 pseudoaneurysm and 1 asymptomatic cerebral thromboembolism present on MRI at discharge). In follow-up, 94% of patients were in sinus rhythm at 3 months. The long journey of RF ablation for AF has reached new heights in improved lesion formation and durability that resulted in incremental success rates of pulmonary vein isolation, linear circumferential being superior to ostial segmental ablations72, from 60-80%58,72,73 freedom of recurrent atrial tachyarrhythmias to high 80-90% with the circumferential ablation guided by newest technologies61,63,65. The safety of AF catheter ablation has improved over the years, a worldwide survey in 2010, that included almost 20,825 procedu-res in 16,309 patients, reported major complications in 4.5% of patients73 (Table).
ONE-SHOT RADIOFREQUENCY ABLATION
Efforts to streamline the ablation procedure, decrease procedural time, increase reproducibility and assure contiguity of the lesions, led to the development of one shot ablation approach. Several tools for radiofrequency ablation became available including multi-electrode and balloon catheters. Multi-electrode ablation catheters have a circular shape with 9 or 10 electrodes, along with multichannel RF generator capable of simultaneously delivering duty-cycled energy, provide the ability to deliver uni-polar or bipolar RF applications or combination of both to multiple electrodes at once. Multiple series have reported the results of ablations using PVAC® (Medtronic Ablation Frontiers, Carlsbad, CA) a first generation, non-irrigated catheter. In Tailored Treatment of Permanent Atrial Fibrillation (TTOP-AF) trial at 6 months, 56% of ablated patients were free from AF, compared to 26% of those treated with antiarrhythmic therapy74. It yielded similar acute, medium and long-term AF ablation success rates as point-by-point ablations, better in paroxysmal AF patients (me-dian 74%, IQR 59–83%) than persistent AF patients (median 55% IQR 47–81%), with significantly lower procedural and fluoroscopy times75-78. However these results were overshadowed by concerns of increased symptomatic (2.9%) and asymptomatic cerebral thromboembolism (38-39%) rates compared to other ablation technologies79-81, and the catheter was not approved in US. The second-generation PVAC GOLD (Medtronic, Minneapolis, MN, USA) catheter was redesigned to mitigate these risks. The platinum-iridium electrodes were replaced with gold, for enhanced thermal conductivity and power output, with greater passive cooling capacity. Other changes in shape included new 20°canted forward shape of circular array, for optimal contact, and reduction of number of electrodes to 9 electrodes, to eliminate electrode interference and reduce the risk of overheating observed between electrodes 1 and 10 (thought to be the possible cause of asymptomatic cerebral thromboembolism)82. In addition, the software upgrade included improved algorithms through the generator for more accurate electrode temperature detection. Gold AF registry included 1071 patients and showed a 12 months success rate of 78% (better in paroxysmal AF 82% than persistent AF 68%) and improved safety, reporting a total of 2.5%82 compared with previously reported PVAC complications of 3.9% 76, and symptomatic ischemic cerebral embolism of 0.3% compared to 1.1-2.9% with PVAC74,76. However, despite updated technology several series still report a concerning high incidence of asymptomatic cerebral embolic events in up to 26% patients on vitamin K antagonists and 33% patients on direct anticoagulants, despite uninterrupted anti-coagulation and high levels of anticoagulation during procedure83. Furthermore, a randomized trial of 70 patients showed a significantly higher incidence of new cerebral lesion on post procedural MRI, 8/35 (23%) of patients ablated with PVAC Gold vs. 2/35 (6%) of patients ablated with ThermoCool (Biosense Webster, Inc. Diamond Bar, CA, USA) catheter84. Following the same development path of point-by-point ablation catheters, an open irrigated multi-electrode catheter was developed: nMARQ™ (Biosense Webster, Inc. Diamond Bar, CA, USA) is an irrigated circular RF catheter visible and integrated with the CARTO system (Biosense Webster, Inc. Diamond Bar, CA, USA). The first in humans experience was in March 2013, and the nMARQ™ catheter was recalled from clinical use in June 2015 due to safety concerns of high silent cerebral lesions (33%), high incidence of esophageal thermal injuries (33%)85 and reporting of three deaths in a small study, of which two were confirmed to be due to esophageal-atrial fistula75,86. The success of ablation at one year in multiple small series was 66-87% freedom of atrial arrhythmias. A new prototype is currently under evaluation75. Of note, a number of the series using multi-electrode catheters reported the need for focal RF ablation “touch-ups” in 2.7-5.7%, to achieve complete pulmonary vein isolation75. The uncertainty of the multielectrode catheters future, with the lessons learned from cryoballoon therapies, radiofrequency balloon based technology was developed for one-shot ablation. To date there are 3 systems available: Heliostar balloon (Biosense Webster, Diamond Bar, CA, USA), Luminize balloon (Boston Scientific, Marlborough, MA, USA), and Globe catheter (Kardium, Burnaby, Canada). The Heliostar compliant balloon has 10 circular oriented, irrigated electrodes, on the surface that can be used for visualization, stimulation, recording and ablation in conjunction with the Carto3 system87. The balloon delivers RF energy directly through the electrodes, forming a continuous circular ablation lesion around the PV ostia. In RADIANCE, the first in human trial, acute success was 100% pulmonary veins isolation using the balloon, with 4.6% acute reconnections after adenosine injection, successfully reisolated with the balloon88. There was only one primary adverse event, right phrenic nerve injury. At 12 months, the freedom from AF was 76% off antiarrhythmic medication87. Currently the Safety and Effectiveness Evaluation of the Multi-Electrode Radiofrequency Balloon Catheter for the Treatment of Symptomatic Paroxysmal Atrial Fibrillation (STELLAR) trial is conducted (Clini-calTrials.gov Identifier: NCT03683030) with expected completion in April 2022. The Luminize balloon has 12 equatorial and 6 distal irrigated electrodes with advantage of direct visualization of the endocardial structures with 4 cameras and LED illumination, for assessment of tissue contact. The AFFICIENT first in-human trial enrolled 18 patients, evaluated the efficacy and safety of the balloon in phase I and showed acute isolation using the balloon in 89% of pulmonary veins with 80% freedom from AF at 6 months89. In phase II, after device enhancements additional 81 patients were enrolled and acute PVI in-creased to 99.4%. There were no device related serious events at 30 days post-procedure. At 12 months, 78.1% of the patients (phase I and II of the trial) were free of atrial arrhythmias90. The Globe is a globular array consisting of 16 ribs with 122 gold-plated flat, non-irrigated electrodes, each of whom perform multiple functions including ablation, measurement of tissue contact and tempe-rature using a sensor positioned directly behind the electrode, recording intracardiac electrograms as well as tissue stimulation. The catheter is capable of generation multiple maps, the FLOW and CONTACT maps providing continue information about contact with atrial tissue and creating anatomy, the VOLTAGE and WAVE maps assessing voltage and electrical activation. Ablation can be performed using up to16-24 electrodes simultaneously with temperatures up to 65°C. The GLOBE AF trial enrolled 60 patients and acute isolation of 99.1% of the veins ablated91. In 34 patients, in whom “single-hot-shot” strategy was considered, ablation was applied with up to 24 electrodes and a temperature set point of up to 65°C, acute PVI was achieved in 100% veins. There were 2 pericardial tamponade related to transseptal puncture and catheter insertion in the left atrium. In 28 patients ablation had to be stopped due to esophageal temperature increase. At 12 months freedom from atrial tachyar-rhyhtmias, off antiarrhythmic medication was 72.3%.
CRYO-ENERGY ABLATION
Cryo-energy ablation produces myocardial tissue damage by direct cellular injury with extreme cooling. There are 3 phases of cellular destruction with cryo energy: 1) the freeze/thaw phase, 2) the hemorrhagic-inflammatory phase, and 3) the replacement fibrosis phase43,92 . In the initial freeze/thaw phase the cryorefrigerant draws heat out of healthy tissue, progressive cooling leading to the formation of ice crystals initially extracellular, below -15°C and then intracellular as temperature falls below -40°C. The crystals cause cellular mechanical disruption, however the predominant mechanism of injury is biochemical, created by extracellular ice crystals hypertonic environment which attracts intracellular water. This leads to increase intracellular saline concentrations with reduction of pH that affects the organelles, leading to irreversible lesions. At the same time microvascular injury occurs. As the thaw occurs, hyperemic vascular response and enlargement of the crystals extends further cellular damage. Following the thaw, inflammation and hemorrhage ensue leading to more cellular destruction via edema and apoptosis. Lastly, the final phase is comprised of tissue infiltration by inflammatory cells and eventual replacement fibrosis over weeks43,92. Several advantages of cryothermal energy have been recognized over RF energy including less patient discomfort, excellent contact by attachment of the catheter to the tissue during freeze, creation of well-demarcated and homogenous lesions, preservation of the tissue structural integrity and the lesions result in minimal endocardial surface disruption, being less thrombogenic92. The original application of cryoablation was in the 1980s in surgical epicardial accessory pathway ablation43. Cryoablation for AF was introduced as adjunctive energy source for Cox-Maze procedures16. Afterwards, the endocardial focal cryo catheters were developed and used in AF ablations as “touch-up” lesion deliveries, usually for posterior antral regions of the pulmonary veins, when RF applications would result in significant increase in esophageal temperature. However, the need for long application time of 3-4 min and freeze-thaw-freeze sequence, point-by point cryoablation was not a viable stand-alone solution for PV isolation. Thus one shot, cryothermal balloon ablation technology was developed, with first system (CryoCath Technologies Inc., Kirkland, Cana-da) consisting of a non-deflectable, over-the-wire 12-F two-lumen catheter with double inner-outer cooling balloons, with 23 mm or 28 mm diameter93. The refrigerant, liquid nitrous oxide, was delivered under pressure from the console into the inner balloon chamber with the 4 jets oriented towards the equatorial belt of the balloon. A liquid-to-gas phase change results in inner balloon cooling temperatures of ≤-80°C. The temperature during cryo application is monitored via a thermocouple located at the inner balloon. The initial study of feasibility on 57 patients took place in 2007 in Netherland and acute isolation using the cryo-balloon (CB) only was achieved in 84% of the veins, the rest requiring touchup using focal cryo catheter93. At 3 months follow-up, using daily ECG recordings, 34 patients (60%) did not have any AF recurrence. There was a signifiant reduction in mean AF burden at 3 months compared to baseline (24 ± 31% baseline to 5 ± 15% at 3 months p < 0.01). The only significant complications related to the catheter were 4 cases (7%) of right phrenic nerve injury (PNI) with cryo application at the right superior pulmonary vein, 2 of them reco-vered during the procedure, one at 3 months and one persisted at 6 months. The first randomized trial comparing cryoballoon ablation to AAD therapy was the Sustained Treatment of Paroxysmal Atrial Fibrillation (STOP-AF) trial, and enrolled 245 patients with paroxysmal AF, who have failed 1.2 antiarrhythmics, with a 2:1 randomization to CB based PV isolation (n=163) versus AAD therapy (n=82)9. Acute PVI by the CB only, was achieved in 91% of participants, and success increased to 98.2% with additional focal cryoablation. A repeat procedure within the 3-month blanking period was performed in 19% of the patients. At 12 months of follow-up, 69.9% of the CB group (114/163) vs. 7.3% of the AAD group (6/82) were free of recurrent AF (p<0.001). An improved, second CB (CB2) generation and a third CB generation (with a shorter tip), has eight jets directed towards the front hemisphere, delivering a more uniform cooling energy to the front of the CB. This upgrade has shown to improve the success rates at 1 year to 84% arrhythmia free survival off antiarrhythmic medication compared to 64-66% with first generation CB (CB1)94,95, with decrease in procedural time, fluoroscopy and increased rate of isolation with first application. Compared to point-by-point radio-frequency PV isolation CB ablation has proven to be non-inferior in FIRE AND ICE trial, which randomized in 1:1 fashion 762 patients to either ablation strategy. Arrhythmia recurrence was seen in 35% of patients treated with CB1 and CB2 and 36% of patients treated with RF ablation (HR 0.96, CI 0.76-1.22, p < 0.001 for non-inferiority)96. The complication rate was similar for both strategies, however with higher PNI in CB arm, seen in 10 (2.7%) patients at discharge, however only 1 (0.3%) patient still had PNI at one year. Long term results of CB2, up to 3 years, were reported in STOP-AF PAS (post approval study), which enrolled 344 patients with paroxysmal AF97. Acute PVI success was 99.3%, using 28 mm CB in 89% of patients, 23 mm CB in 8.4% of patients and both in 2.6% of patients. Additional focal ablation using either cryo or RF ca-theters was necessary in 7.3% of patients. Major procedure related adverse events occurred in 20 patients (5.8%), the most frequent complication being PNI in 11 patients (3.2%), rest being 1 (0.3%) stroke, 2 (0.6%) asymptomatic PV stenosis, 3 (0.9%) hemoptysis and 3 (0.9%) pericardial effusions, one requiring pericardiocentesis. At 36 months PNI persisted in 1 (0.3%) patient. Freedom from atrial arrhythmias recurrence was 79% at one year, 71% at 2 years and 64% at 3 years97. Multiple studies and meta-analyses98–100 showed similar results of CB when compared to standard RF ablation, with shorter procedure time, higher fluoroscopy time and less non-AF atrial tachyarrhythmias98. The CB2 and CF RF catheters have improved the long term ≥12 months outcomes with freedom from atrial tachyarrhyhtmias of 78.1% and 78.2% respectively, compared to first generation technology 57.9% with CB1 and 58.1% RF with non-CF catheters98. Serious complications has been shown to be similar between the two technologies 3.34% for CB2 and 2.94% for CF RF99, with less pericardial effusions and tamponade seen with CB, and PNI almost exclusively seen with CB98-100. Phrenic nerve injury has been recognized to be one of the major Achile’s heel of CB technology. This has been a known complication for other balloon based ablation technologies using different energies, given close proximity of right phrenic nerve and orifice of right sided pulmonary veins101 and wider area of contact and displacement with balloon pressure forward to maintain occlusion. In one series of 100 patients undergoing focal RF ablation102, pace mapping of the phrenic nerve showed capture in 30% of the patients at the levels of anterior orifices of right veins and carina (85% at the carina level, 70% right superior vein and 30% at the right inferior vein). Reported incidence of PNI with point-by-point ablations is 0.17-0.48%102, however with CB technology could vary from 1-17%98,100,101. Stimulation of the phrenic nerve with pacing at the level of superior vena cava during has proven to be a valuable precaution during isolation of the right veins. A number of monitoring strategies of phrenic nerve capture have been employed, either direct tactile sensing of diaphragm contraction strength, direct visualization of diaphragm movement by intracardiac echocardiogram, or diaphragmatic compound motor action potentials (CMAP) recording using 2 surface electrodes at the level of the right diaphragmatic area103,104. Termination of the CB application with diminishing diaphragm contraction or more than 30% decrease in the CMAP signal amplitude showed to be effective in decreasing the incidence of PNI. Although most of recent series show a decrease in PNI inciden-ce to 0-2% in105-108, there are still wide variations with some of the series reporting up to 9% PNI109. Several protocols have evaluated the optimal CB application duration to maximize lesion effectiveness and avoid collateral damages. A single 3 or 4 minutes CB application proved same efficacy at one year as previously reported series, using 2 applications, with 71-82% freedom from AF at one year after single procedure110-112. Two randomized trials showed that shorter or single CB applications are as effective as 3-4 minutes freeze-thaw-freeze applictions106,109 . The CIRCA-DOSE trial106 randomized 346 patients with paroxysmal AF to CB application for 4 minutes, CB application for 2 minutes, each followed by an additional same duration application after PVI, and RF ablation using CF catheter. Freedom at one year from atrial tachyarrhythmia by continuous rhythm monitoring using a loop recorder was 52.2%, 51.7%, and 53.9% with 4-minutes CB application, 2-minutes CB application, and CF-RF, respectively (P=0.87). Freedom from symptomatic atrial tachyarrhythmia at one year was 78.2%, 73.3%, and 79.1% with 4-minutes CB application, 2-minutes CB application, and CF-RF, respectively (P=0.26). AF burden was reduced by a median of 99.9% (interquartile range, 65.3%–100.0%) with 4-minutes CB application, 98.4% (interquartile range, 56.2%–100.0%) with 2-minutes CB application, and 99.3% (interquartile range, 67.8%–100.0%) with CF-RF (P=0.36). Serious complications occurred in 4.6% of patients, with no difference between the treatment arms. There was one esophageal perforation in CF-RF group with pericardial abscess requiring surgical drainage, 3 (1.3%) PNI in the cryoballoon arm, all of which resolved, and 1 (0.9%) stroke occurred in the 4-minute CB arm. The AD-Balloon study109 randomized 110 patients to either additional 3 minutes freeze cycle (AD group) after the freeze that led to PVI or not. Delayed enhancement MRI was performed in 62 patients (28 AD group and 34 non-AD group) to evaluate the lesions. At 1 year freedom from atrial tachyarrhythmias was 87.3% in the AD group and 89.1% in the non-AD group (log-rank test P=0.78). DE-MRI showed no significant difference in the gaps on the PVI lines, 46% in the AD group versus 36% in the non-AD group (P=0.38). Complications occurred in 18 (16%), most of them being PNI in 8 (7%) patients, all resolved by last follow-up, 2 (2%) pericardial effusion/tampona-de, 1 PV stenosis. Although initially approved for ablation of paroxys-mal AF, CB PVI has been an effective ablation strategy for persistent AF as demonstrated by non-randomi-zed series113,114, and a meta-analysis108. More recently, the CB was approved in US for ablation of persistent AF ablation after CRYO4PERSISTENT AF trial107 de-monstrated to be a valid strategy. The single arm prospective trial enrolled 101 patients with persistent AF and 100% AF burden of 7-180 days. At 12 months, 61% of patients were free of atrial tachyarrhythmias with symptomatic improvement in 76% of patients. Major adverse events occurred in 4% of the patients, 2 pseudoaneurysms, one transient ST elevation and one pericardial effusion not requiring intervention, and no PNI107. The improved efficacy and safety of the CB ablation as well as low success rates of antiarrhythmic medication led for consideration of CB PVI as the first treatment strategy for patients with paroxysmal AF. Two recent randomized trials STOP-AF First (US only) with 203 patients and Cryo-FIRST (Europe, Australia and Latin America) with 220 patients randomized to CB PVI or medical treatment were reported as late breaking trials at European Congress of Cardiology, August 29 – September 1, 2020. The freedom from atrial tachyarrhythmias at 12 months was significantly higher, 75% and 82% of patients, in CB arm compared to medically treated patients, 45% and 68%, respectively. POLARx™ Cardiac Cryoablation System (Boston Scientifi c, Marlborough, MA, USA) is a new system using a compliant CB, with promising initial experience in 30 patients presented at Heart Rhythm Scientific Sessions May 8-11, 2019, and showed 100% acute PVI (33% single application), 100% EGM visualization in the veins with no serious adverse events at 30 days115. The Cryoballoon in the Treatment of Symptomatic Drug Refractory Paroxysmal Atrial Fibrillation (FROZEN-AF) Investigational Device Exemption (IDE) study (ClinicalTrials.gov Identifi er: NCT04133168) currently started recruiting patients in June 2020 with an expected completion date in April 2022. The cryoballoon technology has proven to be an excellent alternative strategy for treatment of paro-xysmal and persistent AF with success rates similar to RF ablation, but improved procedural time and lower cardiac complications. The PNI still remains a concerning complication, however this is almost unanimously reversible.
ALTERNATIVE AND NEW ABLATION ENERGIES
In the race for the ideal energy for AF ablation, several other energies have been or are currently in evaluation including heat, LASER energy, high-intensity focused ultrasound (HIFU), liquid nitrogen, pulse field electroporation.
RADIOFREQUENCY HOT BALLOON
The SATAKE HotBalloon ablation system (Toray Industries, Inc., Tokyo, Japan) has outcomes data published from trials conducted mostly in Japan116-118. The system is composed of a balloon that can be inflated up to 26 to 33 mm in diameter with 10 to 20 ml contrast medium and a radiofrequency generator that automatically controls the temperature of the balloon at a preset value (40°C to 70°C) and agitates the fluid inside the balloon. The system uses radiofrequency cur-rent of 1.8 MHz delivered between the coil electrode inside the balloon and the 4 cutaneous electrode patches on the patient’s back to induce capacitive-type heating in the balloon. The thermal energy conducted by the heated balloon, and not direct radiofrequency energy, is used for tissue ablation116. The acute success of pulmonary vein isolation using the balloon only ranged from 80-93%, with medium and long term success rates improving from 59% in the initial cohort116 to 84-93% in subsequent series117,118. As the learning curve improved the initial high rates of pulmonary vein stenosis of 5.2% and phrenic nerve injury of 3.7% im-proved dramatically to none reported in subsequent series.
LASER ABLATION
Lasers use an optical fiber with a radiating fiber tip, in conjunction with a 980-nm wavelength diode, to produce high-energy coherent beams that absorbed by the water in the tissue result in dielectric heating43. Tissue destruction occurs by mechanical damage as well caused by shock waves. The visually guided laser balloon second-generation system (VGLB; Heart-Light; CardioFocus Inc, Marlborough, MA, USA) is comprised of a variable-diameter compliant balloon filled with deuterium oxide, a 2 mm endoscope allowing direct visualization of the PVs and diode delivering laser energy in 30° controllable arcs. The laser energy delivery is titratable, ranging from 5.5 W to 12 W for 20-30 seconds, depending on anterior or posterior wall delivery or presence of blood in the delivery field. Direct visualization allows the operator to ensure complete vein occlusion to avoid delivery of energy to the blood, which could result in thrombus formation, or in the vein, to prevent stenosis.43,89 The first large randomized multicenter trial was The Heart-Light Study comparing safety and efficacy of the second-generation VGLB to RF ablation (non–CF irrigated ablation catheter)119. The study randomized 353 patients with paroxysmal AF in one of the 2 arms. The acute PVI was similar in both groups (99.7% VGLB group and 99.1% in the RF group). At 12 months, freedom from atrial arrhythmias, off antiarrhythmic therapy was similar in the VGLB and RF ablation group (61.1% vs 61.7%, P = 0.003 for noninferiority). As for safety in the VGLB arm a significantly higher rate of diaphragmatic paralysis (3.5% vs 0.6%, P = 0.05) was encountered despite a protocol of phrenic pacing during ablation of the RSPV. A meta-analysis including 17 studies that enrolled 1188 patients, 80% with paroxysmal AF, showed an acute PVI of 98.8% and a 12-month freedom from atrial arrhythmias of 72.9% (74.3% for paroxysmal AF). The most common procedural complication was PNI in 2.6% patients120. The VGLB technology has several advantages including direct visualization of energy delivery, stable position with contiguous lesions, ability to titrate energy at different areas, however one of the concerns is the lack of a safety mechanism at high temperatures, so high energy application could result in crater formation and even tissue perforation43.
HIGH-INTENSITY FOCUSED ULTRASOUND ABLATION
High-intensity focused ultrasound (HIFU), with 20 kHz to 200 MHz range, produces water molecules oscillation in the tissues; the kinetic energy generated is converted to thermal energy that leads to tissue destruction43. The HIFU system (ProRhythm, Ronkonkoma, NY,USA) is comprised of 2 non-compliant balloons: a distal one fluid-filled (24 mm, 27 mm, and 32 mm) with an integrated 9-MHz ultrasound crystal, and a proximal second balloon filled with carbon dioxide. The catheter delivers non-titratable ultrasound energy in a focused ring at about 4 mm distal to the balloon’s surface89. The initial studies121,122 showed a relatively low acute PVI with a high incidence of irreversible phrenic nerve palsy and esophageal injury with a case of fatal atrioesophageal fistula89,123. This led to discontinuation of human trials using HIFU catheter-based ablation. HIFU energy may still offer promise for surgical epicardial ablation, where transmural lesions are unhampered by epicardial fat43.
ULTRALOW TEMPERATURE CRYOABLATION
A novel intelligent Continuous Lesion Ablation System (iCLAS™, Adagio Medical, Laguna Hills, USA) now at its’ third generation uses a 20 electrodes linear cryoablation catheter capable of adopting multiple shapes using different pre-formed stylets, with a distal portion used for mapping and proximal cryo application region (Figure 2). The refrigerant used by the system is liquid Nitrogen able of cooling temperatures to -196°C, with applications of 30-60 seconds for lesion creation. Esophageal protection is assured by continuous circulating saline at 36°C into a compliant intra-esophageal balloon. For the right veins ablation the right phrenic nerve is stimulated with pacing at the superior vena cava level. The initial in humans experience was reported at Heart Rhythm Scientific Sessions in 2018124, when the results of AF ablation in 32 patients were presented. All patients successful underwent PVI, some underwent additionally posterior wall isolation, cavo-tricuspid isthmus ablation and non-PV triggers ablation. At 6 months, 90% of patients were free of AF. Initial safety concerns included 2 PNI, that completely resolved; since introduction of the cryomapping, higher temperature for 30 seconds prior to going into full freeze, PNI was not seen in the last 19 patients. The system has received CE mark in Europe and currently iCLAS™ for Persistent Atrial Fibrillation IDE trial (ClinicalTrials.gov Identifier: NCT04061603) is currently undergoing in several centers in US and Europe. Adagio Medical recently also reported encouraging pre-clinical effectiveness of a Pulse Field Cryoablati-on (PFCA) catheter, combining ultra-low temperature cryo and electroporation.
PULSE FIELD ELECTROPORATION ABLATION
Irreversible electroporation (IRE) is a technology adapted from oncology, where is used for treatment of certain solid cancers125. Application of a high, pulsed electric field (PEF) across the cell increases cell membrane permeability by creation of nano-pores, leading to loss of cell’s homeostasis and death. The PEF is produced by high voltage direct current delivered between 2 or more electrodes126. The components for IRE delivery includes PEF generators, capable to provide high voltage electric fields of 250-3000 V and the catheters capable to deliver the PEF to the tissu-es through multiple channles126,127. Multiple preclinical studies were performed using different, custom developed, protocols typically for IRE a pulse duration of nano to microseconds, usually 100μs (the longer the pulse, the more tissue destruction), 10-90 pulses (the higher number of pulses, the higher the voltage), with a repetition frequency of 1-10 Hz (the higher frequency, the higher the heat generated)126. The attractiveness of this type of energy is the non-thermal mode of cell demise, ultra-rapid delivery (seconds) and most unique feature, tissue selectivity – tissues have specific characteristic threshold of PEF strength that leads to necrosis43,127,128. The cardiomyocytes have one of the lowest thresholds (400V/cm), thus limiting damage of pericardiac structures. Direct application of high voltage PEF to the esophagus resulted in only limited and reversible lesions127. Due to absence of coagulative necrosis, the risk for PV stenosis is minimal, despite transmural lesion demonstration and application direct at the PV ostium126. The first in-human study was reported by Reddy et al.128 with 22 patients undergoing PV isolation, 15 endocardial and 7 epicardial approach. For endocardial ablation PV isolation was 100 % successful in all 57 PVs using a mean of 3.26±0.5 lesions. The epicardial approach was 86% successful (6 of 7 patients) using a mean of two lesions. There were no complications. A new lattice-tip catheter (Sphere-9; Affera, Inc, Watertown, MA), capable to deliver RF and PFA provided by a high-current RF and PF dual-generator, was studied in a multicenter, single arm trial that enrolled 76 patients129. For 40 patients RF was applied at the anterior wall and PFA (RF/PFA) to the posterior wall and in 36 patients only PFA was applied. Acute electrical PVI was achieved in 100% of the veins and conduction block was demonstrated in 99% of the lines performed. A thorough safety evaluation showed no device related complications. One vascular access injury required surgical correction. During ablation at the posterior wall, only low level esophageal heating was seen (<39°C). Esophagogastroduodenoscopy was performed in all the patients at 1.6±1.1 days and showed in PF/PFA group minor mucosal thermal injuries (5.6%) and no lesions in PFA group. Post-procedure brain MRIs were performed in 51of 76 (67%) patients at 1.2±0.6 days, revealing silent cerebral events in 8 of 51 (15.7%) patients, which was asymptomatic. The field of pulse field ablation (PFA) is at the beginnings and despite excellent early results, long term data in efficacy and safety are needed for its validation as a worthwhile ablation technology.
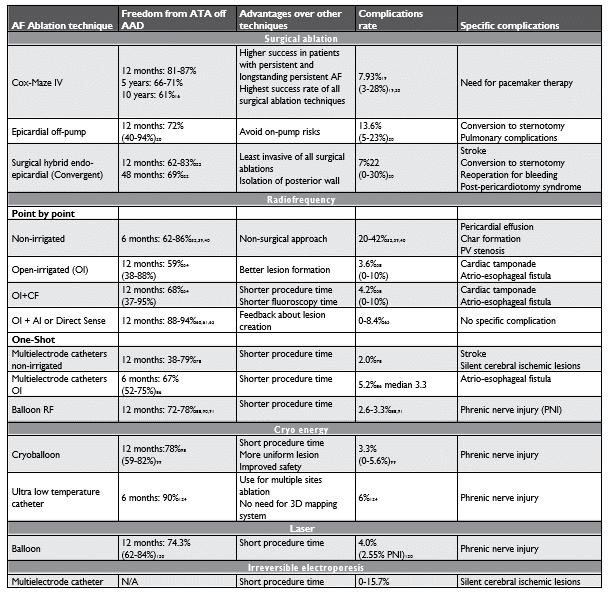
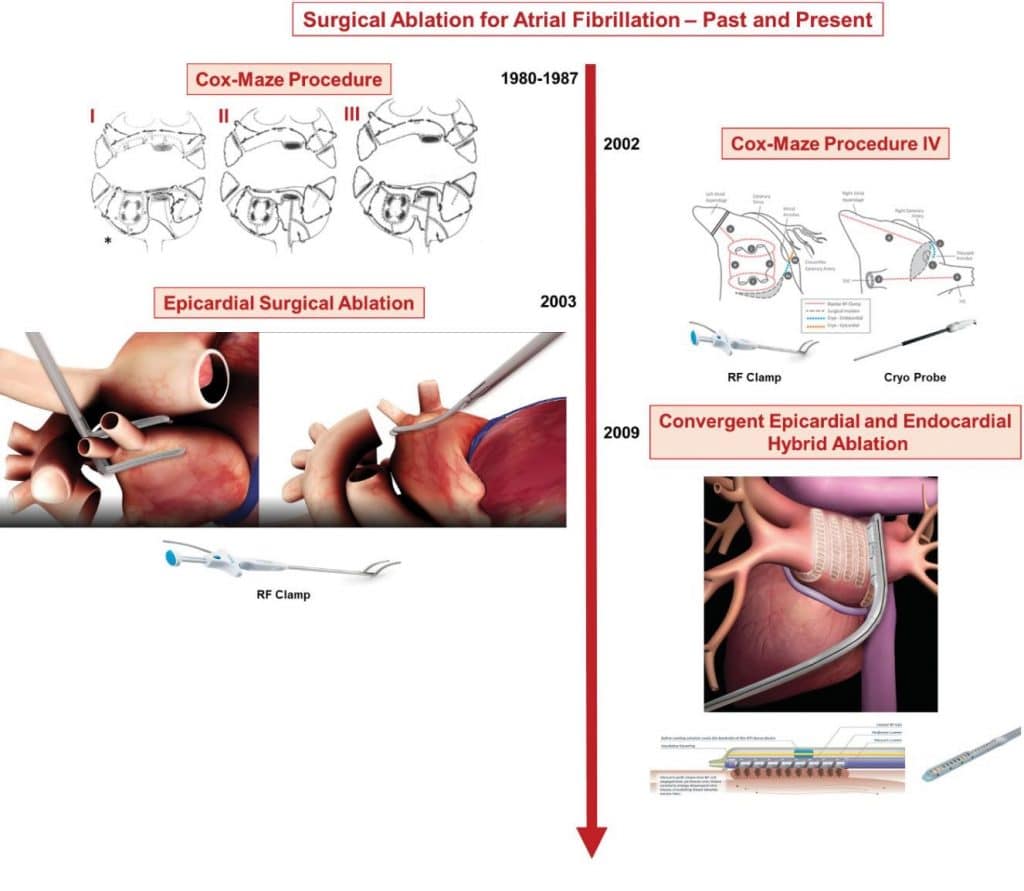
Figure 1. Surgical Ablation for Atrial Fibrillation – Past and Present. * Reproduced with permission from James L Cox et al Seminars in Thoracic and Cardiovascular Surgery 200014; # Reproduced with permission from Robertson et al. Ann Cardiothorac Surg132. The other figures incorporated are courtesy of Atricure Company.
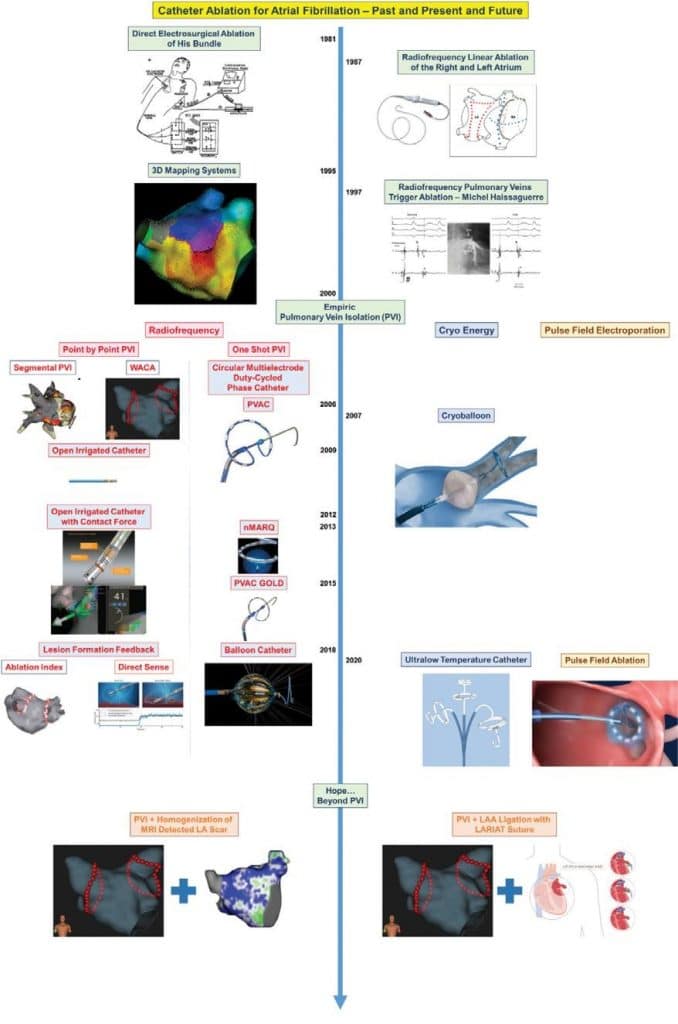
Figure 2. Catheter Ablation for Atrial Fibrillation – Past, Present and Future. * Reproduced with permission from Reproduced with permission from Gal-lagher et al NEJM 198226; # Reproduced with permission from Haissaguerre et al. NEJM 199832; The other figures incorporated are courtesy of Medtronic, Abbott, Boston Scientifi c, Biosense Webster and Adagio Companies.
CONCLUSIONS
The field of AF catheter ablation has evolved at an amazing pace over the past half century, PVI still remaining to date the cornerstone of catheter ablation procedure for both paroxysmal and persistent AF. Despite multiple advances in technologies, the success rate of the ablations is not comparable to the results of ablation for other supraventricular tachycardias. It is still unclear if the way to higher success is a better understanding the pathophysiology at various stages of AF or in developing better tools for PVI. The highest success is achieved in paroxysmal AF, with an effective and durable PVI, likely by addressing the AF PV triggers and neuromodulator inputs. However, once the disease process advances and AF becomes persistent, other factors, besides triggers, are likely involved in maintenance of AF, mainly the atrial substrate remodeling with dilation, scar formation and trigger regions other than PV. Several other ablation techniques, besides PVI, have been employed, including linear ablations, complex fractionated atrial electrograms (CFAE), ablation of non-PV triggers, rotor mapping and ablation with questionable successes. The STAR AF II randomized study130 in patients with persistent AF showed that PVI alone is as good as PVI plus additional linear and/or CFAE ablations. Currently several studies are recruiting or are in follow-up phase evaluating if additional techniques to the PVI as LAA ligation (aMAZE trial)131, posterior wall isolation (iCLAS trial), homogenization of left atrial scar regions detected by MRI (DECAAF II trial) improve the success of ablation for persistent or longstanding persistent AF. The results will be available in the near future, hopefully answering some of these unknowns. The war on AF is not won yet, however many barricades have been conquered, understanding our pitfalls, towards a closer victory (Figure 2).
Conflict of interest: none declared.
References
1. Hindricks G, Potpara T, Dagres N, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collabo-ration with the European Association of Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fi brillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Asso-ciation (EHRA) of the ESC. European Heart Journal. 2020;(ehaa612). doi:10.1093/eurheartj/ehaa612
2. Lippi G, Sanchis-Gomar F, Cervellin G. Global epidemiology of atrial fibrillation: An increasing epidemic and public health chal-lenge. International Journal of Stroke. Published online January 19, 2020:1747493019897870. doi:10.1177/1747493019897870
3. Sieck S. The Economic Impact of Atrial Fibrillation in the US. In: Short Stay Management of Atrial Fibrillation. Contemporary Cardi-ology. Humana Press, Cham; 2016. Accessed September 27, 2020. https://doi.org/10.1007/978-3-319-31386-3_3
4. Velleca M, Costa G, Goldstein L, Bishara M, Boo LM. A Review of the Burden of Atrial Fibrillation: Understanding the Impact of the New Millennium Epidemic across Europe. European Medical Journal. 2019;7(1):110-118.
5. January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS Guide-line for the Management of Patients With Atrial Fibrillation: A Re-port of the American College of Cardiology/American Heart Associ-ation Task Force on Practice Guidelines and the Heart Rhythm Soci-ety. Journal of the American College of Cardiology. 2014;64(21):e1-e76. doi:https://doi.org/10.1016/j.jacc.2014.03.022
6. January Craig T., Wann L. Samuel, Calkins Hugh, et al. 2019 AHA/ ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm So-ciety in Collaboration With the Society of Thoracic Surgeons. Circu-lation. 2019;140(2):e125-e151. doi:10.1161/CIR.0000000000000665
7. Steinberg Jonathan S., O’Connell Heather, Li Shelby, Ziegler Paul D. Thirty-Second Gold Standard Definition of Atrial Fibrillation and Its Relationship With Subsequent Arrhythmia Patterns. Circulation: Ar-rhythmia and Electrophysiology. 2018;11(7):e006274. doi:10.1161/ CIRCEP.118.006274
8. Winkle RA. Atrial fibrillation ablation in patients with transvenous devices: Safety and insights into getting rid of the 30-second rule de-fining procedural success. Journal of Cardiovascular Electrophysiol-ogy. 2019;30(5):688-690. doi:10.1111/jce.13892
9. Packer DL, Kowal RC, Wheelan KR, et al. Cryoballoon Ablation of Pulmonary Veins for Paroxysmal Atrial Fibrillation: First Results of the North American Arctic Front (STOP AF) Pivotal Trial. Jour-nal of the American College of Cardiology. 2013;61(16):1713-1723. doi:10.1016/j.jacc.2012.11.064
10. Wilber DJ, Pappone C, Neuzil P, et al. Comparison of Antiarrhyth-mic Drug Therapy and Radiofrequency Catheter Ablation in Patients With Paroxysmal Atrial Fibrillation: A Randomized Controlled Trial. JAMA. 2010;303(4):333-340. doi:10.1001/jama.2009.2029
11. Wazni OM, Marrouche NF, Martin DO, et al. Radiofrequency Abla-tion vs Antiarrhythmic Drugs as First-line Treatment of Symptomat-ic Atrial FibrillationA Randomized Trial. JAMA. 2005;293(21):2634-2640. doi:10.1001/jama.293.21.2634
12. Pappone C, Augello G, Sala S, et al. A Randomized Trial of Cir-cumferential Pulmonary Vein Ablation Versus Antiarrhythmic Drug Therapy in Paroxysmal Atrial Fibrillation: The APAF Study. Journal of the American College of Cardiology. 2006;48(11):2340-2347. doi:10.1016/j.jacc.2006.08.037
13. Chen C, Zhou X, Zhu M, et al. Catheter ablation versus medical therapy for patients with persistent atrial fibrillation: a system-atic review and meta-analysis of evidence from randomized con-trolled trials. Journal of Interventional Cardiac Electrophysiology. 2018;52(1):9-18. doi:10.1007/s10840-018-0349-8
14. Cox JL, Schuessler RB, Boineau JP. The Development of the Maze Procedure for the Treatment of Atrial Fibrillation. Seminars in Tho-racic and Cardiovascular Surgery. 2000;12(1):2-14. doi:10.1016/ S1043-0679(00)70010-4
15. Henn MC, Lancaster TS, Miller JR, et al. Late outcomes after the Cox maze IV procedure for atrial fi brillation. The Journal of Thoracic and Cardiovascular Surgery. 2015;150(5):1168-1178.e2. doi:10.1016/j. jtcvs.2015.07.102
16. Khiabani AJ, MacGregor RM, Bakir NH, et al. The long-term out-comes and durability of the Cox-Maze IV procedure for atrial fi brilla-tion. The Journal of Thoracic and Cardiovascular Surgery. Published online 2020. doi:https://doi.org/10.1016/j.jtcvs.2020.04.100
17. Badhwar V, Rankin JS, Damiano RJ, et al. The Society of Thorac-ic Surgeons 2017 Clinical Practice Guidelines for the Surgical Treatment of Atrial Fibrillation. The Annals of Thoracic Surgery. 2017;103(1):329-341. doi:10.1016/j.athoracsur.2016.10.076
18. Musharbash FN, Schill MR, Sinn LA, et al. Performance of the Cox-maze IV procedure is associated with improved long-term survival in patients with atrial fibrillation undergoing cardiac surgery. The Jour-nal of Thoracic and Cardiovascular Surgery. 2018;155(1):159-170. doi:10.1016/j.jtcvs.2017.09.095
19. Ad N, Suri RM, Gammie JS, Sheng S, O’Brien SM, Henry L. Sur-gical ablation of atrial fibrillation trends and outcomes in North America. The Journal of Thoracic and Cardiovascular Surgery. 2012; 144(5):1051-1060. doi:10.1016/j.jtcvs.2012.07.065
20. Je HG, Shuman DJ, Ad N. A systematic review of minimally invasive surgical treatment for atrial fibrillation: a comparison of the Cox-Maze procedure, beating-heart epicardial ablation, and the hybrid procedure on safety and efficacy †. European Journal of Cardio-Tho-racic Surgery. 2015;48(4):531-541. doi:10.1093/ejcts/ezu536
21. Osmancik P, Budera P, Talavera D, et al. Five-year outcomes in car-diac surgery patients with atrial fibrillation undergoing concomi-tant surgical ablation versus no ablation. The long-term follow-up of the PRAGUE-12 Study. Heart Rhythm. 2019;16(9):1334-1340. doi:https://doi.org/10.1016/j.hrthm.2019.05.001
22. Wats K, Kiser A, Makati K, et al. The Convergent AF Ablation Pro-cedure: Evolution Of A Multidisciplinary Approach To AF Manage-ment. Arrhythmia & Electrophysiology Review. 2020;9(2):88–96. doi:https://doi.org/10.15420/aer.2019.20
23. Khan Z, Hamandi M, Khan H, DiMaio JM, Evans M. Convergent epi-cardial-endocardial ablation for treatment of long-standing persis-tent atrial fibrillation: A review of literature. Journal of Cardiac Sur-gery. 2020;35(6):1306-1313. doi:10.1111/jocs.14562
24. Weiner MM, Baron EL, Joshi K, et al. Catheter Versus Surgical Ab-lation of Atrial Fibrillation: An Analysis of Outcomes. Journal of Cardiothoracic and Vascular Anesthesia. 2018;32(5):2435-2443. doi:10.1053/j.jvca.2018.01.015
25. Scheinman MM, Morady F, Hess DS, Gonzalez R. Catheter-In-duced Ablation of the Atrioventricular Junction to Control Refrac-tory Supraventricular Arrhythmias. JAMA. 1982;248(7):851-855. doi:10.1001/jama.1982.03330070039027
26. Gallagher JJ, Svenson RH, Kasell JH, et al. Catheter Technique for Closed-Chest Ablation of the Atrioventricular Conduction System. N Engl J Med. 1982;306(4):194-200. doi:10.1056/NEJM 198201283060402
27. Huang SK, Bharati S, Graham AR, Lev M, Marcus FI, Odell RC. Closed chest catheter desiccation of the atrioventricular junction using radiofrequency energy-a new method of catheter ablation. 1987;9(2):349-358. doi:doi: 10.1016/s0735-1097(87)80388-1
28. HAÏSSAGUERRE M, GENCEL L, FISCHER B, et al. Successful Cath-eter Ablation of Atrial Fibrillation. Journal of Cardiovascular Electro-physiology. 1994;5(12):1045-1052. doi:10.1111/j.1540-8167.1994. tb01146.x
29. HAÍSSAGUERRE M, JAÍS P, SHAH DC, et al. Right and Left Atrial Radiofrequency Catheter Therapy of Paroxysmal Atrial Fibrillation. Journal of Cardiovascular Electrophysiology. 1996;7(12):1132-1144. doi:10.1111/j.1540-8167.1996.tb00492.x
30. Gaita Fiorenzo, Riccardi Riccardo, Calò Leonardo, et al. Atrial Map-ping and Radiofrequency Catheter Ablation in Patients With Id-iopathic Atrial Fibrillation. Circulation. 1998;97(21):2136-2145. doi:10.1161/01.CIR.97.21.2136
31. Anselmino M, D’Ascenzo F, Amoroso G, Ferraris F, Gaita F. His-tory of transcatheter atrial fi brillation ablation. Journal of Cardio-vascular Medicine. 2012;13(1). https://journals.lww.com/jcardiovas-cularmedicine/Fulltext/2012/01000/History_of_transcatheter_atri-al_fibrillation.1.aspx
32. Haïssaguerre M, Jaïs P, Shah DC, et al. Spontaneous Initiation of Atrial Fibrillation by Ectopic Beats Originating in the Pulmonary Veins. N Engl J Med. 1998;339(10):659-666. doi:10.1056/NEJM 199809033391003
33. Jai¨s Pierre, Hai¨ssaguerre Michel, Shah Dipen C., et al. A Focal Source of Atrial Fibrillation Treated by Discrete Radiofrequency Ab-lation. Circulation. 1997;95(3):572-576. doi:10.1161/01.CIR.95.3.572
34. Pappone Carlo, Oreto Giuseppe, Lamberti Filippo, et al. Cath-eter Ablation of Paroxysmal Atrial Fibrillation Using a 3D Map-ping System. Circulation. 1999;100(11):1203-1208. doi:10.1161/01. CIR.100.11.1203
35. Lo L-W, Chen S-A. Three-Dimensional Electroanatomic Mapping Systems in Catheter Ablation of Atrial Fibrillation. Circulation Jour-nal. 2010;74(1):18-23. doi:10.1253/circj.CJ-09-0676
36. BULAVA A, HANIS J, EISENBERGER M. Catheter Ablation of Atri-al Fibrillation Using Zero-Fluoroscopy Technique: A Randomized Trial. Pacing and Clinical Electrophysiology. 2015;38(7):797-806. doi:10.1111/pace.12634
37. Lyan E, Tsyganov A, Abdrahmanov A, et al. Nonfluoroscopic cath-eter ablation of paroxysmal atrial fibrillation. Pacing and Clinical Elec-trophysiology. 2018;41(6):611-619. doi:10.1111/pace.13321
38. Doshi R. Atrial Fibrillation Ablation Without Fluoroscopy: Because We Can. The Journal of Innovations in Cardiac Rhythm Manage-ment. 2018;9(11):3391–3394. doi:10.19102/icrm.2018.091103
39. Chen Shih-Ann, Hsieh Ming-Hsiung, Tai Ching-Tai, et al. Initiation of Atrial Fibrillation by Ectopic Beats Originating From the Pulmo-nary Veins. Circulation. 1999;100(18):1879-1886. doi:10.1161/01. CIR.100.18.1879
40. Haïssaguerre Michel, Jaïs Pierre, Shah Dipen C., et al. Electro-physiological End Point for Catheter Ablation of Atrial Fibrilla-tion Initiated From Multiple Pulmonary Venous Foci. Circulation. 2000;101(12):1409-1417. doi:10.1161/01.CIR.101.12.1409
41. Pappone Carlo, Santinelli Vincenzo, Manguso Francesco, et al. Pul-monary Vein Denervation Enhances Long-Term Benefit After Cir-cumferential Ablation for Paroxysmal Atrial Fibrillation. Circulation. 2004;109(3):327-334. doi:10.1161/01.CIR.0000112641.16340.C7
42. Kampaktsis PN, Oikonomou EK, Y. Choi D, Cheung JW. Effi cacy of ganglionated plexi ablation in addition to pulmonary vein isolation for paroxysmal versus persistent atrial fibrillation: a meta-analysis of randomized controlled clinical trials. Journal of Interventional Cardi-ac Electrophysiology. 2017;50(3):253-260. doi:10.1007/s10840-017-0285-z
43. Kowlgi GN, Kapa S. Advances in Atrial Fibrillation Ablation: En-ergy Sources Here to Stay. Cardiac Electrophysiology Clinics. 2020;12(2):167-174. doi:10.1016/j.ccep.2020.02.005
44. HAINES DE, WATSON DD. Tissue Heating During Radiofrequen-cy Catheter Ablation: A Thermodynamic Model and Observations in Isolated Perfused and Superfused Canine Right Ventricular Free Wall. Pacing and Clinical Electrophysiology. 1989;12(6):962-976. doi:10.1111/j.1540-8159.1989.tb05034.x
45. WITTKAMPF FHM, NAKAGAWA H. RF Catheter Ablation: Lessons on Lesions. Pacing and Clinical Electrophysiology. 2006;29(11):1285-1297. doi:10.1111/j.1540-8159.2006.00533.x
46. NATH S, DiMARCO JP, HAINES DE. Basic Aspects of Radiofre-quency Catheter Ablation. Journal of Cardiovascular Electrophysiol-ogy. 1994;5(10):863-876. doi:10.1111/j.1540-8167.1994.tb01125.x
47. Thomas SP, Aggarwal G, Boyd AC, Jin Y, Ross DL. A comparison of open irrigated and non-irrigated tip catheter ablation for pulmonary vein isolation. EP Europace. 2004;6(4):330-335. doi:10.1016/j.eupc. 2004.03.001
48. Neuzil Petr, Reddy Vivek Y., Kautzner Josef, et al. Electrical Recon-nection After Pulmonary Vein Isolation Is Contingent on Contact Force During Initial Treatment. Circulation: Arrhythmia and Electro-physiology. 2013;6(2):327-333. doi:10.1161/CIRCEP.113.000374
49. Reddy Vivek Y., Dukkipati Srinivas R., Neuzil Petr, et al. Random-ized, Controlled Trial of the Safety and Effectiveness of a Contact Force–Sensing Irrigated Catheter for Ablation of Paroxysmal Atrial Fibrillation. Circulation. 2015;132(10):907-915. doi:10.1161/CIRCU-LATIONAHA.114.014092
50. Natale A, Reddy VY, Monir G, et al. Paroxysmal AF Catheter Abla-tion With a Contact Force Sensing Catheter: Results of the Prospec-tive, Multicenter SMART-AF Trial. Journal of the American College of Cardiology. 2014;64(7):647-656. doi:10.1016/j.jacc.2014.04.072
51. Kuck K-H, Reddy VY, Schmidt B, et al. A novel radiofrequency ab-lation catheter using contact force sensing: Toccata study. Heart Rhythm. 2012;9(1):18-23. doi:10.1016/j.hrthm.2011.08.021
52. Reddy VY, Shah D, Kautzner J, et al. The relationship between con-tact force and clinical outcome during radiofrequency catheter ab-lation of atrial fibrillation in the TOCCATA study. Heart Rhythm. 2012;9(11):1789-1795. doi:10.1016/j.hrthm.2012.07.016
53. Kautzner J, Neuzil P, Lambert H, et al. EFFICAS II: optimization of catheter contact force improves outcome of pulmonary vein isola-tion for paroxysmal atrial fibrillation. EP Europace. 2015;17(8):1229-1235. doi:10.1093/europace/euv057
54. Lin H, Chen Y-H, Hou J-W, Lu Z-Y, Xiang Y, Li Y-G. Role of contact force-guided radiofrequency catheter ablation for treatment of atrial fi brillation: A systematic review and meta-analysis. Journal of Car-diovascular Electrophysiology. 2017;28(9):994-1005. doi:10.1111/ jce.13264
55. Lee G, Hunter RJ, Lovell MJ, et al. Use of a contact force-sensing ab-lation catheter with advanced catheter location significantly reduces fl uoroscopy time and radiation dose in catheter ablation of atrial fi-brillation. EP Europace. 2016;18(2):211-218. doi:10.1093/europace/ euv186
56. HUSSEIN AA, BARAKAT AF, SALIBA WI, et al. Persistent Atri-al Fibrillation Ablation With or Without Contact Force Sensing. Journal of Cardiovascular Electrophysiology. 2017;28(5):483-488. doi:10.1111/jce.13179
57. Macle L, Frame D, Gache LM, Monir G, Pollak SJ, Boo LM. Atri-al fibrillation ablation with a spring sensor-irrigated contact force-sensing catheter compared with other ablation catheters: systematic literature review and meta-analysis. BMJ Open. 2019;9(6):e023775. doi:10.1136/bmjopen-2018-023775
58. Virk SA, Ariyaratnam J, Bennett RG, Kumar S. Updated systematic review and meta-analysis of the impact of contact force sensing on the safety and efficacy of atrial fibrillation ablation: discrepancy be-tween observational studies and randomized control trial data. EP Europace. 2019;21(2):239-249. doi:10.1093/europace/euy266
59. Calkins Hugh. Demonstrating the Value of Contact Force Sens-ing. Circulation. 2015;132(10):901-903. doi:10.1161/CIRCULA-TIONAHA.115.018354
60. Hussein Ahmed, Das Moloy, Riva Stefania, et al. Use of Ablation Index-Guided Ablation Results in High Rates of Durable Pulmonary Vein Isolation and Freedom From Arrhythmia in Persistent Atrial Fibrillation Patients. Circulation: Arrhythmia and Electrophysiology. 2018;11(9):e006576. doi:10.1161/CIRCEP.118.006576
61. Phlips T, Taghji P, El Haddad M, et al. Improving procedural and one-year outcome after contact force-guided pulmonary vein isolation: the role of interlesion distance, ablation index, and contact force variability in the ‘CLOSE’-protocol. EP Europace. 2018;20(FI_3):f419-f427. doi:10.1093/europace/eux376
62. Das M, Loveday JJ, Wynn GJ, et al. Ablation index, a novel marker of ablation lesion quality: prediction of pulmonary vein reconnec-tion at repeat electrophysiology study and regional differences in target values. EP Europace. 2017;19(5):775-783. doi:10.1093/euro-pace/euw105
63. Ioannou A, Papageorgiou N, Lim WY, et al. Efficacy and safety of ab-lation index-guided catheter ablation for atrial fibrillation: an updated meta-analysis. EP Europace. 2020;(euaa224). doi:10.1093/europace/ euaa224
64. AVITALL B, HORBAL P, VANCE D, KOBLISH J, KALINSKI A. Maxi-mal Electrogram Attenuation Recorded from Mini Electrodes Em-bedded on 4.5-mm Irrigated and 8-mm Nonirrigated Catheters Sig-nifies Lesion Maturation. Journal of Cardiovascular Electrophysiol-ogy. 2015;26(2):192-202. doi:10.1111/jce.12568
65. Segreti L, De Simone A, Schillaci V, et al. A novel local impedance algorithm to guide effective pulmonary vein isolation in atrial fibril-lation patients: Preliminary experience across different ablation sites from the CHARISMA pilot study. Journal of Cardiovascular Electro-physiology. 2020;31(9):2319-2327. doi:10.1111/jce.14647
66. Martin CA, Martin R, Gajendragadkar PR, et al. First clinical use of novel ablation catheter incorporating local impedance data. Jour-nal of Cardiovascular Electrophysiology. 2018;29(9):1197-1206. doi: 10.1111/jce.13654
67. Sulkin Matthew S., Laughner Jacob I., Hilbert Sebastian, et al. Novel Measure of Local Impedance Predicts Catheter–Tissue Contact and Lesion Formation. Circulation: Arrhythmia and Electrophysiology. 2018;11(4):e005831. doi:10.1161/CIRCEP.117.005831
68. Shin DG, Ahn J, Han S-J, Lim HE. Efficacy of high-power and short-duration ablation in patients with atrial fibrillation: a prospective randomized controlled trial. EP Europace. 2020;22(10):1495-1501. doi:10.1093/europace/euaa144
69. Qiu J, Wang Y, Wang DW, Hu M, Chen G. Update on high-power short-duration ablation for pulmonary vein isolation. Journal of Car-diovascular Electrophysiology. 2020;31(9):2499-2508. doi:10.1111/ jce.14649
70. Castrejón-Castrejón S, Martínez Cossiani M, Ortega Molina M, et al. Feasibility and safety of pulmonary vein isolation by high-power short-duration radiofrequency application: short-term results of the POWER-FAST PILOT study. Journal of Interventional Cardiac Elec-trophysiology. 2020;57(1):57-65. doi:10.1007/s10840-019-00645-5
71. Reddy VY, Grimaldi M, De Potter T, et al. Pulmonary Vein Isolation With Very High Power, Short Duration, Temperature-Controlled Lesions: The QDOT-FAST Trial. JACC: Clinical Electrophysiology. 2019;5(7):778-786. doi:10.1016/j.jacep.2019.04.009
72. Proietti Riccardo, Santangeli Pasquale, Di Biase Luigi, et al. Compara-tive Effectiveness of Wide Antral Versus Ostial Pulmonary Vein Iso-lation. Circulation: Arrhythmia and Electrophysiology. 2014;7(1):39-45. doi:10.1161/CIRCEP.113.000922
73. Cappato Riccardo, Calkins Hugh, Chen Shih-Ann, et al. Updated Worldwide Survey on the Methods, Efficacy, and Safety of Cath-eter Ablation for Human Atrial Fibrillation. Circulation: Arrhyth-mia and Electrophysiology. 2010;3(1):32-38. doi:10.1161/CIR-CEP.109.859116
74. Hummel J, Michaud G, Hoyt R, et al. Phased RF ablation in persistent atrial fibrillation. Heart Rhythm. 2014;11(2):202-209. doi:10.1016/j. hrthm.2013.11.009
75. Laish-Farkash A, Suleiman M. Laish-Farkash A, M. Comparison of the Efficacy of PVAC® and nMARQTM for paroxysmal atrial fibrillation. JAFIB. 2017;9(6):1550. doi:10.4022/jafib.1550
76. Scharf C, Ng GA, Wieczorek M, et al. European survey on efficacy and safety of duty-cycled radiofrequency ablation for atrial fibrilla-tion. EP Europace. 2012;14(12):1700-1707. doi:10.1093/europace/ eus188
77. De Greef Y, Buysschaert I, Schwagten B, Stockman D, Tavernier R, Duytschaever M. Duty-cycled multi-electrode radiofrequency vs. conventional irrigated point-by-point radiofrequency ablation for recurrent atrial fibrillation: comparative 3-year data. EP Europace. 2014;16(6):820-825. doi:10.1093/europace/eut398
78. Andrade JG, Dubuc M, Rivard L, et al. Efficacy and safety of atrial fi-brillation ablation with phased radiofrequency energy and multielec-trode catheters. Heart Rhythm. 2012;9(2):289-296. doi:10.1016/j. hrthm.2011.09.009
79. Wasmer K, Foraita P, Leitz P, et al. Safety profile of multielectrode-phased radiofrequency pulmonary vein ablation catheter and irri-gated radiofrequency catheter. EP Europace. 2016;18(1):78-84. doi: 10.1093/europace/euv046
80. Boersma Lucas V., van der Voort Pepijn, Debruyne Pilippe, et al. Mul-tielectrode Pulmonary Vein Isolation Versus Single Tip Wide Area Catheter Ablation for Paroxysmal Atrial Fibrillation. Circulation: Ar-rhythmia and Electrophysiology. 2016;9(4):e003151. doi:10.1161/ CIRCEP.115.003151
81. Herrera Siklódy C, Deneke T, Hocini M, et al. Incidence of Asymp-tomatic Intracranial Embolic Events After Pulmonary Vein Isolation: Comparison of Different Atrial Fibrillation Ablation Technologies in a Multicenter Study. Journal of the American College of Cardiology. 2011;58(7):681-688. doi:10.1016/j.jacc.2011.04.010
82. Boersma L, Koźluk E, Maglia G, et al. Paroxysmal and persistent atrial fi brillation ablation outcomes with the pulmonary vein ablation cath-eter GOLD duty-cycled phased radiofrequency ablation catheter: quality of life and 12-month efficacy results from the GOLD Atrial Fibrillation Registry. EP Europace. 2020;22(6):888-896. doi:10.1093/ europace/euaa042
83. Wieczorek M, Bogossian H, Bandorski D, Hoeltgen R. Uninter-rupted use of direct oral anticoagulants versus vitamin K antagonists for catheter ablation of atrial fi brillation with PVAC gold: incidence of silent cerebral microembolic events. Journal of Interventional Cardiac Electrophysiology. Published online September 19, 2020. doi:10.1007/s10840-020-00863-2
84. Keçe F, Bruggemans EF, de Riva M, et al. Incidence and Clinical Signif-icance of Cerebral Embolism During Atrial Fibrillation Ablation With Duty-Cycled Phased-Radiofrequency Versus Cooled-Radiofrequen-cy: A Randomized Controlled Trial. JACC: Clinical Electrophysiol-ogy. 2019;5(3):318-326. doi:10.1016/j.jacep.2018.11.008
85. DENEKE T, SCHADE A, MÜLLER P, et al. Acute Safety and Efficacy of a Novel Multipolar Irrigated Radiofrequency Ablation Catheter for Pulmonary Vein Isolation. Journal of Cardiovascular Electrophysi-ology. 2014;25(4):339-345. doi:10.1111/jce.12316
86. Vurma M, Dang L, Brunner-La Rocca H-P, et al. Safety and efficacy of the nMARQ catheter for paroxysmal and persistent atrial fi brillation. EP Europace. 2016;18(8):1164-1169. doi:10.1093/europace/euw048
87. Dhillon GS, Honarbakhsh S, Di Monaco A, et al. Use of a multi-elec-trode radiofrequency balloon catheter to achieve pulmonary vein isolation in patients with paroxysmal atrial fi brillation: 12-Month out-comes of the RADIANCE study. Journal of Cardiovascular Electro-physiology. 2020;31(6):1259-1269. doi:10.1111/jce.14476
88. Reddy Vivek Y., Schilling Richard, Grimaldi Massimo, et al. Pulmonary Vein Isolation With a Novel Multielectrode Radiofrequency Balloon Catheter That Allows Directionally Tailored Energy Delivery. Cir-culation: Arrhythmia and Electrophysiology. 2019;12(12):e007541. doi:10.1161/CIRCEP.119.007541
89. Bhardwaj R, Neuzil P, Reddy VY, Dukkipati SR. Balloon-Based Ab-lation Technologies. Cardiac Electrophysiology Clinics. 2020;12(2): 175-185.
90. Keller DM. Most Patients Free of AF 1 Year After Visually-Guided RF Ablation. Published July 29, 2020. Accessed October 28, 2020. https://www.medscape.com/viewarticle/934817
91. Kottkamp H, Hindricks G, Pönisch C, et al. Global multielectrode contact-mapping plus ablation with a single catheter in patients with atrial fibrillation: Global AF study. Journal of Cardiovascular Electro-physiology. 2019;30(11):2248-2255. doi:10.1111/jce.14172
92. ANDRADE JG, DUBUC M, GUERRA PG, et al. The Biophys-ics and Biomechanics of Cryoballoon Ablation. Pacing and Clinical Electrophysiology. 2012;35(9):1162-1168. doi:10.1111/j.1540-8159. 2012.03436.x
93. Van Belle Y, Janse P, Rivero-Ayerza MJ, et al. Pulmonary vein iso-lation using an occluding cryoballoon for circumferential ablation: feasibility, complications, and short-term outcome. European Heart Journal. 2007;28(18):2231-2237. doi:10.1093/eurheartj/ehm227
94. GIOVANNI GD, WAUTERS K, CHIERCHIA G-B, et al. One-Year Follow-Up After Single Procedure Cryoballoon Ablation: A Compar-ison Between the First and Second Generation Balloon. Journal of Cardiovascular Electrophysiology. 2014;25(8):834-839. doi:10.1111/ jce.12409
95. FÜRNKRANZ A, BORDIGNON S, DUGO D, et al. Improved 1-Year Clinical Success Rate of Pulmonary Vein Isolation with the Second-Generation Cryoballoon in Patients with Paroxys-mal Atrial Fibrillation. Journal of Cardiovascular Electrophysiology. 2014;25(8):840-844. doi:10.1111/jce.12417
96. Kuck K-H, Brugada J, Fürnkranz A, et al. Cryoballoon or Radiofre-quency Ablation for Paroxysmal Atrial Fibrillation. N Engl J Med. 2016;374(23):2235-2245. doi:10.1056/NEJMoa1602014
97. Knight BP, Novak PG, Sangrigoli R, et al. Long-Term Outcomes Af-ter Ablation for Paroxysmal Atrial Fibrillation Using the Second-Generation Cryoballoon: Final Results From STOP AF Post-Ap-proval Study. JACC: Clinical Electrophysiology. 2019;5(3):306-314. doi:10.1016/j.jacep.2018.11.006
98. CARDOSO R, MENDIRICHAGA R, FERNANDES G, et al. Cryo-balloon versus Radiofrequency Catheter Ablation in Atrial Fibrilla-tion: A Meta-Analysis. Journal of Cardiovascular Electrophysiology. 2016;27(10):1151-1159. doi:10.1111/jce.13047
99. Wang Y, Wang W, Yao J, Chen L, Yi S. Second-generation cryo-balloon vs. contact-force sensing radiofrequency catheter ablation in atrial fibrillation: a meta-analysis of randomized controlled trials. Journal of Interventional Cardiac Electrophysiology. Published online October 11, 2020. doi:10.1007/s10840-020-00893-w
100. Nirav Patel*, Krunalkumar Patel, Abhishek Shenoy, William L. Bak-er, Amgad N. Makaryus and Nabil El-Sherif. Cryoballoon Abla-tion for the Treatment of Atrial Fibrillation: A Meta-analysis. Cur-rent Cardiology Reviews. 2019;15(3):230-238. doi:10.2174/157340 3X15666181212102419
101. ANTZ M, CHUN KJ, OUYANG F, KUCK K-H. Ablation of Atrial Fibrillation in Humans Using a Balloon-Based Ablation System: Iden-tification of the Site of Phrenic Nerve Damage Using Pacing Maneuvers and CARTO. Journal of Cardiovascular Electrophysiology. 2006;17(11):1242-1245. doi:10.1111/j.1540-8167.2006.00589.x
102. Yong Ji S, Dewire J, Barcelon B, et al. Phrenic Nerve Injury: An Un-derrecognized and Potentially Preventable Complication of Pul-monary Vein Isolation Using a Wide-Area Circumferential Ab-lation Approach. Journal of Cardiovascular Electrophysiology. 2013;24(10):1086-1091. doi:10.1111/jce.12210
103. Franceschi Frédéric, Koutbi Linda, Gitenay Edouard, et al. Electro-myographic Monitoring for Prevention of Phrenic Nerve Palsy in Second-Generation Cryoballoon Procedures. Circulation: Arrhythmia and Electrophysiology. 2015;8(2):303-307. doi:10.1161/CIR-CEP.115.002734
104. SHARMA PS, PADALA SK, THOMPSON JJ, GUNDA S, KONERU JN, ELLENBOGEN KA. Factors Influencing Diaphragmatic Com-pound Motor Action Potentials During Cryoballoon Ablation for Atrial Fibrillation. Journal of Cardiovascular Electrophysiology. 2016; 27(12):1384-1389. doi:10.1111/jce.13082
105. Anwar O, Gunawardene MA, Dickow J, et al. Contemporary anal-ysis of phrenic nerve injuries following cryoballoon-based pulmo-nary vein isolation: A single-centre experience with the systematic use of compound motor action potential monitoring. PLOS ONE. 2020;15(6):e0235132. doi:10.1371/journal.pone.0235132
106. Andrade Jason G., Champagne Jean, Dubuc Marc, et al. Cryobal-loon or Radiofrequency Ablation for Atrial Fibrillation Assessed by Continuous Monitoring. Circulation. 2019;140(22):1779-1788. doi:10.1161/CIRCULATIONAHA.119.042622
107. Boveda S, Metzner A, Nguyen DQ, et al. Single-Procedure Out-comes and Quality-of-Life Improvement 12 Months Post-Cryobal-loon Ablation in Persistent Atrial Fibrillation: Results From the Mul-ticenter CRYO4PERSISTENT AF Trial. JACC: Clinical Electrophysi-ology. 2018;4(11):1440-1447. doi:10.1016/j.jacep.2018.07.007
108. Omran H, Gutleben K-J, Molatta S, et al. Second generation cryo-balloon ablation for persistent atrial fibrillation: an updated meta-analysis. Clinical Research in Cardiology. 2018;107(2):182-192. doi: 10.1007/s00392-017-1171-5
109. Miyamoto Koji, Doi Atsushi, Hasegawa Kanae, et al. Multicenter Study of the Validity of Additional Freeze Cycles for Cryobal-loon Ablation in Patients With Paroxysmal Atrial Fibrillation. Cir-culation: Arrhythmia and Electrophysiology. 2019;12(1):e006989. doi:10.1161/CIRCEP.118.006989
110. MIYAZAKI S, HACHIYA H, NAKAMURA H, et al. Pulmonary Vein Isolation Using a Second-Generation Cryoballoon in Patients With Paroxysmal Atrial Fibrillation: One-Year Outcome Using a Single Big-Balloon 3-Minute Freeze Technique. Journal of Cardiovascular Electrophysiology. 2016;27(12):1375-1380. doi:10.1111/jce.13078
111. Wissner E, Heeger C-H, Grahn H, et al. One-year clinical suc-cess of a ‘no-bonus’ freeze protocol using the second-generation 28 mm cryoballoon for pulmonary vein isolation. EP Europace. 2015;17(8):1236-1240. doi:10.1093/europace/euv024
112. Ciconte G, de Asmundis C, Sieira J, et al. Single 3-minute freeze for second-generation cryoballoon ablation: One-year follow-up af-ter pulmonary vein isolation. Heart Rhythm. 2015;12(4):673-680. doi:10.1016/j.hrthm.2014.12.026
113. Tondo C, Iacopino S, Pieragnoli P, et al. Pulmonary vein isolation cryoablation for patients with persistent and long-standing persis-tent atrial fibrillation: Clinical outcomes from the real-world mul-ticenter observational project. Heart Rhythm. 2018;15(3):363-368. doi:10.1016/j.hrthm.2017.10.038
114. Perego GB, Iacopino S, Molon G, et al. Cryoablation for paroxys-mal and persistent AF in patients with structural heart disease and preserved ejection fraction: Clinical outcomes from 1STOP, a mul-ticenter observational project. Journal of Cardiology. 2019;74(1):19-26. doi:10.1016/j.jjcc.2019.02.015
115. Anic A, Lever N, Martin A, et al. S-AB26-05 First in Human Experi-ence: Procedural Safety and Efficacy of a Novel Cryoballoon Pulmo-nary Vein Ablation System. In: Heart Rhythm Journal. Vol 16, Issue 5. ; 2019:S65.
116. Sohara H, Ohe T, Okumura K, et al. HotBalloon Ablation of the Pulmonary Veins for Paroxysmal AF: A Multicenter Randomized Tri-al in Japan. Journal of the American College of Cardiology. 2016; 68(25):2747-2757. doi:10.1016/j.jacc.2016.10.037
117. Yamasaki H, Aonuma K, Shinoda Y, et al. Initial Result of Antrum Pulmonary Vein Isolation Using the Radiofrequency Hot-Balloon Catheter With Single-Shot Technique. JACC: Clinical Electrophysi-ology. 2019;5(3):354-363. doi:10.1016/j.jacep.2019.01.017
118. Yamasaki H, Aonuma K, Shinoda Y, et al. Lesion durability after an-tral pulmonary vein isolation using a radiofrequency hot balloon catheter. Journal of Interventional Cardiac Electrophysiology. Pub-lished online September 16, 2020. doi:10.1007/s10840-020-00857-0
119. Dukkipati SR, Cuoco F, Kutinsky I, et al. Pulmonary Vein Isolation Using the Visually Guided Laser Balloon: A Prospective, Multicenter, and Randomized Comparison to Standard Radiofrequency Ablation. Journal of the American College of Cardiology. 2015;66(12):1350-1360. doi:10.1016/j.jacc.2015.07.036
120. Reynolds MR, Zheng Q, Doros G. Laser balloon ablation for AF: A systematic review and meta-analysis. Journal of Cardiovascular Elec-trophysiology. 2018;29(10):1363-1370. doi:10.1111/jce.13698
121. Schmidt B, Antz M, Ernst S, et al. Pulmonary vein isolation by high-intensity focused ultrasound: First-in-man study with a steerable balloon catheter. Heart Rhythm. 2007;4(5):575-584. doi:10.1016/j. hrthm.2007.01.017
122. Metzner A, Chun KRJ, Neven K, et al. Long-term clinical outcome following pulmonary vein isolation with high-intensity focused ul-trasound balloon catheters in patients with paroxysmal atrial fibrillation. EP Europace. 2010;12(2):188-193. doi:10.1093/europace/ eup416
123. Borchert B, Lawrenz T, Hansky B, Stellbrink C. Lethal atrioesopha-geal fistula after pulmonary vein isolation using high-intensity focused ultrasound (HIFU). Heart Rhythm. 2008;5(1):145-148. doi:10.1016/j. hrthm.2007.08.023
124. De Potter T, Boersma L, Babkin A, Mazor M, Cox J. Novel Linear Cryoablation Catheter to Treat Atrial Fibrillation. In: Heart Rhythm Journal. Vol 15, No 5. ; 2018:S132.
125. Rubinsky B, Onik G, Mikus P. Irreversible Electroporation: A New Ablation Modality — Clinical Implications. Technol Cancer Res Treat. 2007;6(1):37-48. doi:10.1177/153303460700600106
126. Maor E, Sugrue A, Witt C, et al. Pulsed electric fields for cardi-ac ablation and beyond: A state-of-the-art review. Heart Rhythm. 2019;16(7):1112-1120. doi:https://doi.org/10.1016/j.hrthm.2019.01.12
127. Sugrue A, Vaidya V, Witt C, et al. Irreversible electroporation for catheter-based cardiac ablation: a systematic review of the preclini-cal experience. Journal of Interventional Cardiac Electrophysiology. 2019;55(3):251-265. doi:10.1007/s10840-019-00574-3
128. Reddy VY, Koruth J, Jais P, et al. Ablation of Atrial Fibrillation With Pulsed Electric Fields: An Ultra-Rapid, Tissue-Selective Modality for Cardiac Ablation. JACC: Clinical Electrophysiology. 2018;4(8):987-995. doi:10.1016/j.jacep.2018.04.005
129. Reddy Vivek Y., Anter Elad, Rackauskas Gediminas, et al. Lattice-Tip Focal Ablation Catheter That Toggles Between Radiofrequency and Pulsed Field Energy to Treat Atrial Fibrillation. Circulation: Ar-rhythmia and Electrophysiology. 2020;13(6):e008718. doi:10.1161/ CIRCEP.120.008718
130. Verma A, Jiang C, Betts TR, et al. Approaches to Catheter Ablation for Persistent Atrial Fibrillation. N Engl J Med. 2015;372(19):1812-1822. doi:10.1056/NEJMoa1408288
131. Lee RJ, Lakkireddy D, Mittal S, et al. Percutaneous alternative to the Maze procedure for the treatment of persistent or long-stand-ing persistent atrial fibrillation (aMAZE trial): Rationale and design. American Heart Journal. 2015;170(6):1184-1194. doi:10.1016/j. ahj.2015.09.019
132. Robertson JO, Saint LL, Leidenfrost J, Damiano Jr RJ. Illustrated tech-niques for performing the Cox-Maze IV procedure through a right mini-thoracotomy. Annals of Cardiothoracic Surgery; Vol 3, No 1 (January 2014): Atrial fibrillation surgery. Published online 2014. Ac-cessed January 1, 2014. http://www.annalscts.com/article/view/3246.
 This work is licensed under a
This work is licensed under a