Elias J. Gialafos1,2, Theodore G. Papaioannou*3, Elissavet Andreadou1, Spyridon Katsanos4, Elias Tsougos2, A. Dimitrakopoulos1, Panagiotis Kokotis1, Kostas Kilindireas1, Dimitrios Tousoulis3, Maria Anagnostouli1
1 Eginition Hospital, Medical School, National and Kapodistrian University of Athens, Greece; and
2 Second Department of Cardiology, Henry Dunant Hospital Center, Athens, Greece.
3 First Department of Cardiology, Biomedical Engineering Unit, Hippokration Hospital, Medical School, National and Kapodistrian University of Athens, Greece.
4 Second Department of Cardiology, Attikon Hospital, Medical School, National and Kapodistrian University of Athens, Greece.
Abstract: Objectives – Multiple Sclerosis (MS) has been linked with the presence of coronary artery disease including Acute Coronary Syndrome with Normal Coronary Arteries (ACSNCA), but limited data exist regarding the underlying mechanisms. We aimed to explore potential classic risk factors and novel vascular biomarkers related to ACSNCA in MS patients. Methods – One hundred six patients with MS were enrolled. Two groups were defined according to the presence and/or history of ACSNCA. The presence of predisposing factors such as hypertension, diabetes, hyperlipidemia, smoking, obesity and family history was reported. Arterial stiffness and aortic wave reflections were also assessed by pulse wave velocity (PWV) and augmentation index, respectively, calculated by a validated, brachial cuff-based automated oscillometric device. Multiple logistic regression models were constructed to determine the strongest parameters related with the pre-sence of ACSNCA. Results – Eleven patients with episodes of ACSNCA (Group-A) and 95 MS patients free of ACSNCA (Group-B) were identifi ed. Among blood pressure parameters (peripheral and aortic), arterial stiffness and wave reflections, PWV had the strongest independent association with ACSNCA (enter-method). PWV values above 7 m/sec were related with the history of ACSNCA independently from age, diabetes and blood pressure. When all hemodynamic, arterial and classic risk factors were examined in a step-wise (backward) model diabetes and hyperlipidemia were the strongest inde-pendent factors associated with ACSNCA’s presence. Conclusions – In MS patients, diabetes and hyperlipidemia represent the strongest, independent parameters related with the presence of ACSNCA. Arterial stiffness seems to play an underlying role in the development of ACSNCA which merits further investigation.
Keywords: arteries, pulse wave velocity, wave refl ections, cardiovascular risk, autoimmune disease.
INTRODUCTION
Patients with Acute Coronary Syndrome with Nor-mal Coronary Arteries (ACSNCA) is now recognized as an important subgroup of acute coronary syndro-mes1,2. The exact prevalence of ACSNCA is unknown; previous case series reported prevalence between 0.4% and 12%3. The pathophysiology of ACSNCA is not well comprehended and several mechanisms have been suggested including coronary spasm, coronary thrombosis, platelet dysfunction and inflammation.
Multiple Sclerosis (MS) is the most common neuro-logical disorder affecting young people with progres-sive disability. It is characterized by immune-mediated chronic inflammation that can lead to destruction of the myelin sheath around the axons of the central ner-vous system4. There is also evidence relating ACSN-CA and MS and cases of vasospastic angina after the initial diagnosis of MS but the exact mechanisms and triggering factors are unknown5,6.
ARTEMIS study (ARTerial and hEmodynamic proper-ties of patients with MultIple Sclerosis) is a prospective study aiming to explore the cardiovascular (CV) risk of MS patients. In the present observational, cross-sectional study, we present data regarding clinical and novel non-invasive hemodynamic and vascular factors related to ACSNCA.
METHODS
Study design and population
The main inclusion criterion for this study was the retrospective identification of patients with ACSNCA based on coronary arteriography and according to the current guidelines7, in a large registry of MS patients in a tertiary University hospital. Overall, 11 stable pa-tients were found from the database in the outpatient clinic for the CV assessment of our hospital who for-med Group-A. These patients had undergone coro-nary arteriogram during the hospitalization for ACS. We randomly selected from the ARTEMIS registry, with an 1:10 ratio, 110 MS patients free of ACSNCA (Group-B). The diagnosis of MS was based on clini-cal history and neurological examination supported by Magnetic Resonance Imaging of the brain and the spinal cord as well as cerebrospinal fl uid analysis. Two months without any significant new symptoms and/ or signs was defined as a stable phase. Patients having obstructive coronary artery disease, systolic heart fa-ilure (ejection fraction <50%), arrhythmias and/or in antiarrhythmic medication were excluded. Also, the presence of other autoimmune disease was an exclu-sion criterion.
All patients underwent an interview with a cardi-ologist and their medical history was documented in detail, based also in the review of medical files and information from hospital discharge letters. Patients’ neurologic and cardiologic data including Expanded Disability Status Scale (EDSS) were carefully evaluated for demographic characteristics, co-morbid conditi-ons, symptoms of MS, medications and response to medication. Assessment of central hemodynamics and arterial stiffness was performed by the use of a brachi-al cuff-based automated sphygmomanometer.
Standard Protocol Approvals, Registrations, and Patient Consents
The study was approved by the Ethical/Scientific Com-mittee of the Hospital and all participants provided written informed consent according to the declaration of Helsinki.
Measurement of hemodynamic and arterial parameters
Brachial and aortic blood pressures (BP) were assessed after at least 10 min of rest in the sitting position un-der controlled room temperature (22-25 °C). Measu-rements were performed using a commercial, brachial cuff-based automated oscillometric device (Mobil-O-Graph NG, IEM, Stolberg, Germany), with FDA and CE approval for brachial BP measurement. The BP detection unit of the apparatus is validated according to the British Hypertension Society (BHS) protocol and European Society of Hypertension (ESH) International Protocol8,9.
The same device was used for the estimation of aortic BP as previously described10,11. The device is working based on the following principles and tech-nique. At first the device measures the brachial systo-lic and diastolic BP and after that, the cuff which is equipped with a high fidelity pressure sensor, is re-inflated at the level of the diastolic BP for approxi-mately 10 seconds. The sensor records continuously brachial pressure waves which are calibrated using the mean and the brachial diastolic BP values. Aor-tic pulse waveform is then computationally derived by using a generalized transfer function which is imple-mented in the ARCSolver algorithm as previously de-scribed12,13. Arterial stiffness was also assessed by the Mobil-O-Graph system by the ARCSolver algorithm which provides an indirect estimate of PWV through a mathematical model which takes into account several parameters from pulse wave analysis and wave sepa-ration analysis. More specifically, for the computation of PWV, the ARCSolver method utilizes pulse wave analysis and wave separation analysis which are com-bined in a proprietary mathematical model. The major determinants of the model are age, central pressure, and aortic characteristic impedance, but not timing of brachial supra systolic wave reflections14,15. This appa-ratus and technique have been validated previously using invasive and non-invasive data12,16-18.
In Figure 1, mathematically derived aortic pressu-re waveforms (also decomposed to their forward and backward traveling wave components) are illustrated for an MS patient (Figure 1-a) with increased arterial stiffness (above normal PWV) who presented a CAD event and for a MS patient (Figure 2-a) with lower ar-terial stiffness (normal PWV) without any CAD event.
Statistics
Quantitative and categorical data are presented as mean ± standard deviation and as percentages respec-tively. Mann-Whitney test was performed to evaluate differences of continuous variables between MS pati-ents with and without ACSNCA, whereas Chi-Square test or Fischer exact test, as appropriate, were used to assess the respective differences in categorical vari-ables. The discriminatory ability of CV parameters to detect the presence of ACSNCA was evaluated using Receiver Operating Curve (ROC) analysis. Step-wise multiple logistic regression models were construc-ted to determine the stronger parameters related with ACSNCA. Statistical significance was defined as p<0.05. Statistical analysis was performed using SPSS 23 (IBM Corp, Armonk, NY, USA).
RESULTS
Out of the 110 patients without ACSNCA, ninety-five fulfi lled all the study criteria. The descriptive characte-ristics of the study population are reported in Table 1. All patients were under disease modifying medications, including glatiramer acetate (n=16), interferon beta 1b (n=6), mitoxandrone (n=21), natalizumab (n=26), in-terferon beta-1a (n=28), and fi ngolimod (n=9).
We found that both groups were comparable concerning gender, height and weight. However, the patients in group A were older than those in group B (47.9±15.1 vs 38.0±11.8 yrs, p=0.029), had higher prevalence of hypertension (54.5% vs 2.1%, p<0.001), diabetes (27.3% vs 1.1%, p=0.003) and hyperlipide-mia (54.5% vs 9.6%, p=0.001). Both groups had simi-lar profile regarding smoking status (p=0.172), family history of CV disease (p=0.199) and disease severity (EDSS, p=0.434).
Hemodynamic and arterial properties for the total population as well as for the two subgroups of MS patients are reported in Table 2. MS patients with ACSNCA had significantly higher values of brachial and aortic diastolic BP, mean arterial pressure and PWV compared to those without ACSNCA (Table 2).
ROC analysis showed that PWV has a significant ability to detect MS patients with ACSNCA (area un-der curve 0.727, p=0.014). More specifically, PWV greater than 7 m/sec has 72.7% sensitivity and 73.7% specificity to discriminate MS patients with ACSNCA from those without CV events (Figure 2). In the mul-tivariate logistic regression analysis (step-wise) PWV was inserted in the model as a binary independent variable (0 for subjects with PWV <7 m/sec and 1 for those with PWV ≥7 m/sec). Among PWV levels, hyperlipidemia, age, diabetes, mean BP (or diastolic BP) and diabetes, we found that diabetes (p=0.03) and hyperlipidemia (p=0.04) were the strongest indepen-dent factors associated with ACSNCA’s presence.
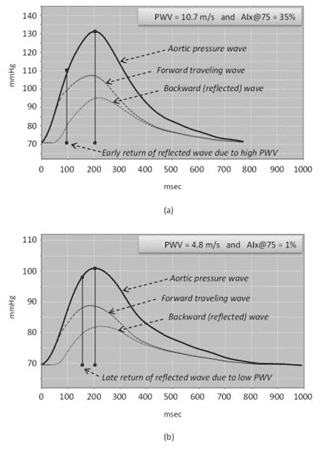
Figure 1. Mathematically derived aortic pressure waveform: (a) for a mul-tiple sclerosis (MS) patient with increased pulse wave velocity (PWV) who presented a cardiovascular event and (b) for an MS patient with low PWV without any cardiovascular event. In case (a), the early return of reflected wave, due to the increased arterial stiffness, caused a greater augmentation of aortic systolic pressure compared to the patient of case (b), where a late return of reflected wave is observed during systole resulting to a small augmentation of aortic systolic pressure (1%).
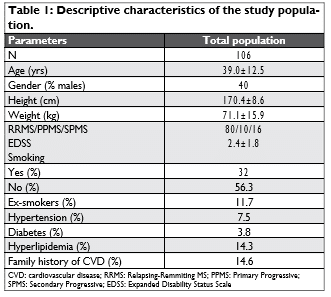
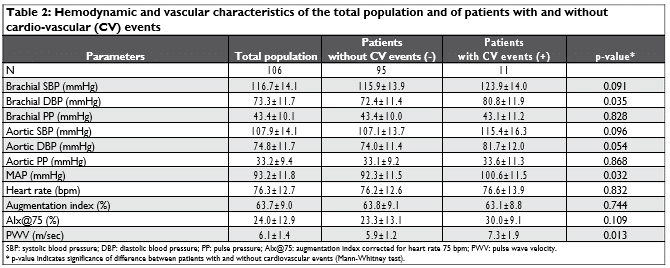
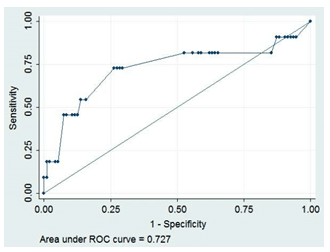
Figure 2. Receiver operator curve plot illustrating the ability of pulse wave velocity to detect MS patients with cardiovascular events.
DISCUSSION
We demonstrated, to the best of our knowledge for the fi rst time, that diabetes mellitus and hyperlipidemia are the strongest predictors of ACSNCA in patients with multiple sclerosis. Among the hemodynamic and vascular indices, PWV was the stronger independent determinant of ACSNCA above peripheral and cen-tral blood pressure parameters and wave reflection indices.
Pathophysiological mechanisms relating multiple sclerosis with cardiovascular disease. Recently, it was shown that MS patients have an in-creased incidence and prevalence for cardiac, cere-brovascular and peripheral vascular disease especially within the first years after a first-time MS diagnosis19,20. Although this risk is less than 5%, it is still signifi cantly higher compared to the general population20.
Multiple Sclerosis and CV disease share common mechanisms like immune dysregulation and inflamma-tory processes that might lead to increased arterial stiffness. Presence of predisposing factors for coro-nary artery disease in patients with MS, such as arte-rial hypertension, diabetes mellitus, smoking, lack of exercise might explain the increased CV risk. Moreo-ver, molecular and/or genetic abnormalities like lower vitamin D levels, higher plasma levels of homocysteine, altered thrombogenic factors, oxidative stress or even CV autonomic dysfunction due to the demyelination process per se might aggravate endothelial dysfunction which represents a step towards atherosclerosis. Our findings showed that diabetes and hyperlipidemia, pos-sibly via these mechanisms, were independently asso-ciated with the presence of ACSNCA in MS patients.
Another interesting finding in our study was the appearance of normal coronary arteries after cardi-ac catheterization, in patients with MS who presen-ted symptoms of acute coronary syndrome. It is also of interest that according to our current knowledge these patients present an increased prevalence of cardiovascular risk factors. A possible explanation to this phenomenon is the appearance of a coronary mi-crovascular spasm under stressful events triggered by an interaction of the demyelinating lesions with cen-tral nervous system, autonomic nervous system and immune system21.
Concerning arterial stiffness, it is well established that it is an independent risk factor for CV risk and mortality22. Based on several published data from pro-spective studies and clinical trials, PWV measurement has been proposed by the guidelines for the manage-ment of arterial hypertension of the European Society of Hypertension/European Society of Cardiology as a tool for the assessment of subclinical target organ damage23. An increased arterial stiffness has been observed in MS patients compared to controls24. In another study, arterial compliance was also found to be significantly compromised in young individuals with MS, compared with age-matched controls, but not for older individu-als25. However, there are no data regarding the asso-ciation of arterial stiffness with ACSNCA to support our fi ndings.
The results of the present study provide initial evidence that PWV is related with ACSNCA in MS patients. More specifically PWV greater than 7 m/sec yielded a 72.7% sensitivity and 73.7% specificity to discriminate MS patients with ACSNCA from those without events. More importantly PWV was still sig-nifi cantly related with ACSNCA regardless from BP parameters (central and peripheral) and wave reflec-tions (data not shown). However, when all parame-ters were examined in a step-wise logistic regression analysis, diabetes and hyperlipidemia were found to be independently associated with the presents of ACSN-CA in MS patients.
Limitations
It should be acknowledged that these preliminary re-sults were derived from a small number of MS patients with ACSNCA. The small sample size and the low number of CV events did not allow the adjustment of the results for all potential confounding factors such as medication and co-morbidities. A prospective study design examining the predictive value of arterial stiff-ness in MS for the development of future CV events would provide stronger evidence. However, this is the first study exploring the relation of PWV and CAD events in this population. Nonetheless, the ARTEMIS study is ongoing with prospective data being continuo-usly collected. Although the new treatment modalities have increased the life expectancy and the quality of life of MS patients, the use of glucocorticoids, interfe-ron and mitoxandrone may also increase the risk of CV disease for MS patients. Unfortunately biomarkers related to inflammatory status of the patients were not measured, although this type of drug therapy exerts potent anti-inflammatory effects.
CONCLUSIONS
In patients with multiple sclerosis, diabetes and hyper-lididemia are independently associated with the pre-sence of ACSNCA. Arterial stiffness seems to play an underlying role in the development of ACSNCA that merits further investigation.
Conflict of interest: TGP received equipment for research purposes by I.E.M. GmbH (Stolberg, Ger-many). The authors declare that they have no other confl ict of interest.
REFERENCES
1. Saba L, Fellini F, De Filippo M. Diagnostic value of contrast-enhanced cardiac magnetic resonance in patients with acute coronary syndrome with normal coronary arteries. Jpn J Radiol. 2015;33:410-417
2. Chacon-Hernandez N, San Miguel-Cervera D, Vilar-Herrero JV, Rumiz-Gonzalez E, Berenguer-Jofresa A, Morell-Cabedo S. Acute coronary syndrome in patients with normal coronary arteries: An optical coherence tomography study. Rev Esp Cardiol (Engl Ed). 2015; 68:533-535
3. Daniel M, Ekenback C, Agewall S, Brolin EB, Caidahl K, Cederlund K, Collste O, Eurenius L, Frick M, Younis-Hassan S, Henareh L, Jernberg T, Malmqvist K, Spaak J, Sorensson P, Hofman-Bang C, Tornvall P. Risk factors and markers for acute myocardial infarction with angiographically normal coronary arteries. Am J Cardiol. 2015;116:838-844
4. Kurtzke JF. Multiple sclerosis in time and space–geographic clues to cause. J Neurovirol. 2000;6 Suppl 2:S134-140
5. Lalouschek W, Muller C, Gamper G, Weissel M, Turetschek K. Myocardial ischemia with normal coronary arteries associated with thoracic myelitis. N Engl J Med. 1997;337:1920
6. Joing S, Casey R, Forgosh L. Prinzmetal variant angina associated with multiple sclerosis. J Am Board Fam Pract. 2004;17:71-73
7. Agewall S, Beltrame JF, Reynolds HR, Niessner A, Rosano G, Caforio AL, De Caterina R, Zimarino M, Roffi M, Kjeldsen K, Atar D, Kaski JC, Sechtem U, Tornvall P, Pharmacotherapy WGoC. Esc working group position paper on myocardial infarction with non-obstructive coronary arteries. Eur Heart J. 2017;38:143-153
8. Franssen PM, Imholz BP. Evaluation of the mobil-o-graph new generation abpm device using the esh criteria. Blood Press Monit. 2010;15:229-231
9. Wei W, Tolle M, Zidek W, van der Giet M. Validation of the mobilograph: 24 h-blood pressure measurement device. Blood Press Monit. 2010;15:225-228
10. Papaioannou TG, Argyris A, Protogerou AD, Vrachatis D, Nasothimiou EG, Sfikakis PP, Stergiou GS, Stefanadis CI. Non-invasive 24 hour ambulatory monitoring of aortic wave reflection and arterial stiffness by a novel oscillometric device: The first feasibility and reproducibility study. Int J Cardiol. 2013;169:57-61
11. Protogerou AD, Argyris A, Nasothimiou E, Vrachatis D, Papaioannou TG, Tzamouranis D, Blacher J, Safar ME, Sfikakis P, Stergiou GS. Feasibility and reproducibility of noninvasive 24-h ambulatory aortic blood pressure monitoring with a brachial cuff-based oscillometric device. Am J Hypertens. 2012;25:876-882
12. Hametner B, Parragh S, Mayer C, Weber T, Van Bortel L, De Buyzere M, Segers P, Rietzschel E, Wassertheurer S. Assessment of model based (input) impedance, pulse wave velocity, and wave refl ection in the asklepios cohort. PLoS One. 2015;10:e0141656
13. Wassertheurer S, Mayer C, Breitenecker F. Modeling arterial and left ventricular coupling for non-invasive measurements. Simul Model Pract Th. 2008;16:988-997
14. Luzardo L, Lujambio I, Sottolano M, da Rosa A, Thijs L, Noboa O, Staessen JA, Boggia J. 24-h ambulatory recording of aortic pulse wave velocity and central systolic augmentation: A feasibility study. Hypertens Res. 2012;35:980-987
15. Aissopou EK, Argyris AA, Nasothimiou EG, Konstantonis GD, Tampakis K, Tentolouris N, Papathanassiou M, Theodossiadis PG, Papaioannou TG, Stehouwer CD, Sfikakis PP, Protogerou AD. Ambulatory aortic stiffness is associated with narrow retinal arteriolar caliber in hypertensives: The safar study. Am J Hypertens. 2016;29:626-633
16. Hametner B, Wassertheurer S, Kropf J, Mayer C, Holzinger A, Eber B, Weber T. Wave reflection quantification based on pressure wave forms alone–methods, comparison, and clinical covariates. Comput Methods Programs Biomed. 2013;109:250-259
17. Hametner B, Wassertheurer S, Kropf J, Mayer C, Eber B, Weber T. Oscillometric estimation of aortic pulse wave velocity: Comparison with intra-aortic catheter measurements. Blood Press Monit. 2013;18:173-176
18. Papaioannou TG, Karageorgopoulou TD, Sergentanis TN, Protogerou AD, Psaltopoulou T, Sharman JE, Weber T, Blacher J, Daskalopoulou SS, Wassertheurer S, Khir AW, Vlachopoulos C, Ster-
giopulos N, Stefanadis C, Nichols WW, Tousoulis D. Accuracy of commercial devices and methods for noninvasive estimation of aortic systolic blood pressure a systematic review and meta-analysis of invasive validation studies. J Hypertens. 2016;34:1237-1248
19. Christiansen CF. Risk of vascular disease in patients with multiple sclerosis: A review. Neurol Res. 2012;34:746-753
20. Marrie RA, Reider N, Cohen J, Stuve O, Trojano M, Cutter G, Reingold S, Sorensen PS. A systematic review of the incidence and prevalence of cardiac, cerebrovascular, and peripheral vascular disease in multiple sclerosis. Mult Scler. 2015;21:318-331
21. Habek M. Immune and autonomic nervous system interactions in multiple sclerosis: Clinical implications. Clin Auton Res. 2019;29:267- 275
22. Vlachopoulos C, Aznaouridis K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: A systematic review and meta-analysis. J Am Coll Cardiol. 2010;55:1318- 1327
23. Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G, Grassi G, Heagerty AM, Kjeldsen SE, Laurent S, Narkiewicz K, Ruilope L, Rynkiewicz A, Schmieder RE, Boudier HA, Zanchetti A, Vahanian A, Camm J, De Caterina R, Dean V, Dickstein K, Filippatos G, Funck-Brentano C, Hellemans I, Kristensen SD, McGregor K, Sechtem U, Silber S, Tendera M, Widimsky P, Zamorano JL, Erdine S, Kiowski W, Agabiti-Rosei E, Ambrosioni E, Lindholm LH, Viigimaa M, Adamopoulos S, Agabiti-Rosei E, Ambrosioni E, Bertomeu V, Clement D, Erdine S, Farsang C, Gaita D, Lip G, Mallion JM, Manolis AJ, Nilsson PM, O’Brien E, Ponikowski P, Redon J, Ruschitzka F, Tamargo J, van Zwieten P, Waeber B, Williams B, Management of Arterial Hypertension of the European Society of H, European Society of C. 2007 guidelines for the management of arterial hypertension: The task force for the management of arterial hypertension of the european society of hypertension (esh) and of the european society of cardiology (esc). J Hypertens. 2007;25:1105-1187
24. Ranadive SM, Yan H, Weikert M, Lane AD, Linden MA, Baynard T, Motl RW, Fernhall B. Vascular dysfunction and physical activity in multiple sclerosis. Med Sci Sports Exerc. 2012;44:238-243
25. Fjeldstad C, Frederiksen C, Fjeldstad AS, Bemben M, Pardo G. Arterial compliance in multiple sclerosis: A pilot study. Angiology. 2010; 61:31-36.
 This work is licensed under a
This work is licensed under a